Research News and Market Data on TSQ
Digital Represents 55% of 2025 Total Net Revenue and 56% of 2025 Total Segment Profit
Reduced Debt by $23 Million Since the February 2025 Refinancing
PURCHASE, N.Y., March 16, 2026 (GLOBE NEWSWIRE) — Townsquare Media, Inc. (NYSE: TSQ) (“Townsquare”, the “Company,” “we,” “us” or “our”) announced today its financial results for the fourth quarter and year ended December 31, 2025.
“I am pleased to share that Townsquare’s fourth quarter and year end results met our previously issued net revenue and Adjusted EBITDA guidance, reflecting our team’s hard work in the current environment. We are proud that the execution of our Digital First Local Media strategy allowed us to deliver excellent results for our clients, while also outperforming competitors and gaining market share,” commented Bill Wilson, Chief Executive Officer of Townsquare Media, Inc. “In 2025, net revenue decreased -2.8% year-over-year excluding political, and -5.2% in total, and Adjusted EBITDA decreased -3.0% year-over-year excluding political, and -12.2% in total. Importantly, due to our strong expense management, Adjusted EBITDA margins excluding political were constant year-over-year, despite revenue declines. In addition, our full year net loss improved by $1.2 million year-over-year, to a net loss of $9.8 million.”
Mr. Wilson continued, “In 2025, Townsquare’s Digital business, which now represents 55% of the Company’s total net revenue and 56% of our total Segment Profit, was buoyed by the consistent strength of our differentiated programmatic digital advertising offering, including the success of our Media Partnership division (which we’re proud to announce has expanded from six to eleven partners in 2026), and the direct sales of our local owned and operated digital properties. Both of these digital revenue streams increased by +9% year-over-year in 2025, offsetting the significant short-term headwinds of declining search engine referral traffic we experienced in 2025, and leading to overall 2025 Digital Advertising revenue growth of +1.6% as compared to the prior year. I’m also pleased to share that we are starting to see early signs of digital audience stabilization, and are optimistic the negative remnant revenue headwinds presented by the year-over-year decline in search engine referral traffic will abate in the second half of 2026. In fact to start 2026, the traffic to our local mobile apps and websites grew sequentially month-over-month with both January and February online audience being larger than December, and thus our local remnant revenue in January and February was more than we generated in any month of Q4. I would also like to highlight the strong profit performance of Townsquare Interactive in 2025, which delivered Segment Profit growth of +17.4% year-over-year (+$3.7 million), and a Segment Profit margin of 34%, representing the highest Segment Profit margin in Townsquare Interactive’s 14-year history.”
“One of the largest benefits of our business model is significant cash generation. In 2025, despite having meaningfully higher interest expense obligations, we generated $30.6 million of cash flow from operations over the course of the year. We utilized this cash flow first and foremost to invest in our local business through organic, internal investments that support our revenue and profit growth, particularly our digital growth engine. In addition, we repaid $22.6 million of debt following our February 2025 refinancing through both voluntary and mandatory debt amortization, and made $13.2 million of dividend payments. Due to management’s and the Board of Director’s ongoing confidence in our business, the underlying strength of our digital advertising differentiation, and our consistent, strong free cash flow characteristics, which we believe is not reflected in our stock price to date, we plan to maintain the dividend at its current rate, despite the high implied dividend yield. Looking forward, we remain confident in our ability to build shareholder value for our investors through long-term net revenue, Adjusted EBITDA and cash flow growth, net leverage reduction, and future dividend payments,” concluded Mr. Wilson.
The Company announced today that its Board of Directors approved a quarterly cash dividend of $0.20 per share. The dividend will be payable on May 4, 2026 to shareholders of record as of the close of business on April 27, 2026. As of the last closing price, this reflects a dividend yield of approximately 11%.
Segment Reporting
We have three reportable operating segments, Digital Advertising, Subscription Digital Marketing Solutions, and Broadcast Advertising. The Digital Advertising segment, marketed externally as Townsquare Ignite, includes digital advertising on our digital programmatic advertising platform and our owned and operated digital properties, and our first party data digital management platform. The Subscription Digital Marketing Solutions segment includes our subscription digital marketing solutions business, Townsquare Interactive. The Broadcast Advertising segment includes our local, regional, and national advertising products and solutions delivered via terrestrial radio broadcast, and other miscellaneous revenue that is associated with our broadcast advertising platform. The remainder of our business is reported in the Other category, which includes our live events business.
Fourth Quarter Results*
- As compared to the fourth quarter of 2024:
- Net revenue decreased 9.6%, and 4.5% excluding political
- Net income decreased $29.8 million from net income of $25.0 million to a net loss of $4.8 million
- Adjusted EBITDA decreased 30.9%, and 17.0% excluding political
- Total Digital net revenue decreased 2.4%
- Digital Advertising net revenue decreased 1.0%
- Subscription Digital Marketing Solutions (“Townsquare Interactive”) net revenue decreased 5.6%
- Total Digital Segment Profit decreased 14.8%
- Digital Advertising Segment Profit decreased 28.0%
- Subscription Digital Marketing Solutions Segment Profit increased 12.0%
- Broadcast Advertising net revenue decreased 17.8%, and 7.9% excluding political
- Net loss per diluted share was $0.32 and Adjusted Net Income per diluted share was $0.05
- Repaid $5.9 million of our Senior Secured Credit Facility, including $3.0 million of the Revolver and $2.9 million of Term Loan
Full Year Results*
- As compared to the year ended December 31, 2024:
- Net revenue decreased 5.2%, and 2.8% excluding political
- Net loss decreased $1.2 million from a net loss of $10.9 million to a net loss of $9.8 million
- Adjusted EBITDA decreased 12.2%, and 3.0% excluding political
- Total Digital net revenue increased 0.9%
- Digital Advertising net revenue increased 1.6%
- Subscription Digital Marketing Solutions net revenue decreased 0.7%
- Total Digital Segment Profit decreased 1.7%
- Digital Advertising Segment Profit decreased 11.7%
- Subscription Digital Marketing Solutions Segment Profit increased 17.4%
- Broadcast Advertising net revenue decreased 12.6%, and 8.0% excluding political
- Entered into a five-year $490 million Credit Agreement, including a $470 million Senior Secured Term Loan Facility and a $20 million Revolving Credit Facility
- Redeemed all of the Company’s outstanding 2026 Senior Secured Notes of $467.4 million
- Reduced outstanding debt by $22.6 million since the February 2025 refinancing, including $14.6 million of our Term Loan and an $8.0 million net reduction of the outstanding Revolving Credit Facility
*See below for discussion of non-GAAP measures.
Guidance
For the first quarter of 2026, net revenue is expected to be between $96 million and $98 million, and Adjusted EBITDA is expected to be between $16 million and $17 million.
For the full year 2026, net revenue is expected to be between $420 million and $440 million, and Adjusted EBITDA is expected to be between $87 million and $93 million.
Quarter Ended December 31, 2025 Compared to the Quarter Ended December 31, 2024
Net Revenue
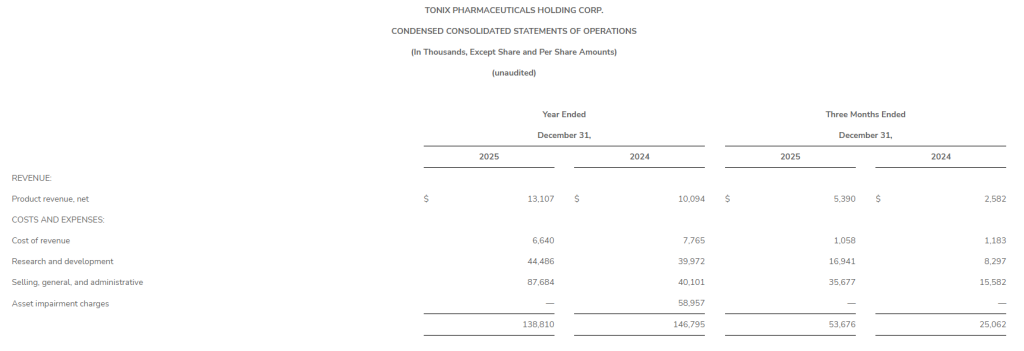
Net revenue for the three months ended December 31, 2025 decreased $11.3 million, or 9.6%, to $106.5 million as compared to $117.8 million in the same period in 2024. Broadcast Advertising net revenue decreased $9.9 million, or 17.8%, due to decreases in the purchases of advertising by our clients and decreases in political revenue. Subscription Digital Marketing Solutions net revenue decreased $1.1 million or 5.6%, due to reduced sales velocity as a result of lower sales headcount, and Digital Advertising net revenue decreased $0.4 million, or 1.0%, primarily due to decreases in remnant digital advertising revenue.
Excluding political revenue of $0.9 million and $7.2 million for the three months ended December 31, 2025 and 2024, respectively, net revenue decreased $5.0 million, or 4.5%, to $105.6 million, Broadcast Advertising net revenue decreased $3.9 million, or 7.9%, to $45.0 million, and Digital Advertising net revenue decreased $0.1 million, or 0.2%, to $41.5 million.
Net (Loss) Income
For the three months ended December 31, 2025, we reported net loss of $4.8 million, a decrease of $29.8 million as compared to net income of $25.0 million in the same period last year. The decrease was due to a $15.1 million increase in the provision for income taxes, a $11.3 million decrease in net revenue and a $3.6 million increase in interest expense. Adjusted Net Income decreased $9.5 million to $0.8 million, as compared to $10.3 million for the same period last year.
Adjusted EBITDA
Adjusted EBITDA for the three months ended December 31, 2025 decreased $9.6 million, or 30.9%, to $21.5 million, as compared to $31.2 million in the same period last year. Adjusted EBITDA (Excluding Political) decreased $4.3 million, or 17.0%, to $20.8 million, as compared to $25.1 million in the same period last year.
Year Ended December 31, 2025 Compared to the Year Ended December 31,2024
Net Revenue
Net revenue for the year ended December 31, 2025 decreased $23.6 million, or 5.2%, to $427.4 million as compared to $451.0 million in the same period in 2024. Broadcast Advertising net revenue decreased $26.5 million, or 12.6%, due to decreases in the purchases of advertising by our clients and decreases in political revenue, and Subscription Digital Marketing Solutions net revenue decreased $0.5 million, or 0.7% due to reduced sales velocity as a result of lower sales headcount. These decreases were partially offset by an increase in Digital Advertising net revenue of $2.6 million, or 1.6%, and an increase in Other net revenue of $0.8 million, or 11.8%.
Excluding political revenue of $2.2 million and $13.4 million for the year ended December 31, 2025 and 2024, respectively, net revenue decreased $12.3 million, or 2.8% to $425.2 million, Broadcast Advertising net revenue decreased $15.8 million, or 8.0%, to $181.5 million, and Digital Advertising net revenue increased $3.1 million, or 2.0%, to $160.9 million.
Net Loss
For the year ended December 31, 2025, we reported net loss of $9.8 million, an improvement of $1.2 million as compared to $10.9 million in the same period last year. The decrease in net loss was due to a $28.8 million decrease in non-cash impairment charges, an $8.5 million decrease in direct operating expenses, an $8.1 million increase in gain on sale and retirement of assets and a $3.4 million decrease in stock-based compensation. These amounts were partially offset by the $23.6 million decrease in net revenue discussed above, an $11.7 million increase in interest expense, a $6.7 million increase in transaction and business realignment cost, and a $5.1 million increase in other (expense) income. Adjusted Net Income decreased $14.8 million to $4.4 million as compared to $19.2 million for the same period last year.
Adjusted EBITDA
Adjusted EBITDA for the year ended December 31, 2025 decreased $12.3 million, or 12.2% to $88.1 million, as compared to $100.4 million in the same period last year. Adjusted EBITDA (Excluding Political) decreased $2.7 million, or 3.0%, to $86.3 million, as compared to $89.0 million in the same period last year.
Liquidity and Capital Resources
As of December 31, 2025, we had a total of $4.8 million of cash and cash equivalents and $457.4 million of outstanding indebtedness, representing 5.19x and 5.14x gross and net leverage, respectively, based on Adjusted EBITDA for the year ended December 31, 2025, of $88.1 million.
The table below presents a summary, as of March 9, 2026, of our outstanding common stock (net of treasury shares).
| Security | Number Outstanding | Description | ||
| Class A common stock | 15,789,817 | One vote per share. | ||
| Class B common stock | 815,296 | 10 votes per share.1 | ||
| Class C common stock | 500,000 | No votes.1 | ||
| Total | 17,105,113 | |||
| 1 Each share converts into one share of Class A common stock upon transfer or at the option of the holder, subject to certain conditions, including compliance with FCC rules. | ||||
Conference Call
Townsquare Media, Inc. will host a conference call to discuss certain fourth quarter 2025 financial results and 2026 guidance on Monday, March 16, 2026 at 8:00 a.m. Eastern Time. The conference call dial-in number is 1-800-717-1738 (U.S. & Canada) or 1-646-307-1865 (International) and the conference ID is “Townsquare.” A live webcast of the conference call will also be available on the investor relations page of the Company’s website at www.townsquaremedia.com.
A replay of the conference call will be available through March 23, 2026. To access the replay, please dial 1-844-512-2921 (U.S. and Canada) or 1-412-317-6671 (International) and enter confirmation code 1134751. A web-based archive of the conference call will also be available at the above website.
About Townsquare Media, Inc.
Townsquare is a community-focused digital and broadcast media and digital marketing solutions company principally focused outside the top 50 markets in the U.S.Townsquare Ignite, our robust digital advertising division, specializes in helping businesses of all sizes connect with their target audience through data-driven, results based strategies, by utilizing a) our proprietary digital programmatic advertising technology stack with an in-house demand and data management platform and b) our owned and operated portfolio of more than 400 local news and entertainment websites and mobile apps along with a network of leading national music and entertainment brands, collecting valuable first party data. Townsquare Interactive, our subscription digital marketing services business, partners with SMBs to help manage their digital presence by providing a SAAS business management platform, website design, creation and hosting, search engine optimization and other digital services. And through our portfolio of local radio stations strategically situated outside the Top 50 markets in the United States, we provide effective advertising solutions for our clients and relevant local content for our audiences. For more information, please visit www.townsquaremedia.com, www.townsquareinteractive.com and www.townsquareignite.com.
Forward-Looking Statements
Except for the historical information contained in this press release, the matters addressed are forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements often discuss our current expectations and projections relating to our financial condition, results of operations, plans, objectives, future performance and business. You can identify forward-looking statements by the fact that they do not relate strictly to historical or current facts. These statements may include words such as “aim,” “anticipate,” “estimate,” “expect,” “forecast,” “outlook,” “potential,” “project,” “projection,” “plan,” “intend,” “seek,” “believe,” “may,” “could,” “would,” “will,” “should,” “can,” “can have,” “likely,” the negatives thereof and other words and terms. Actual events or results may differ materially from the results anticipated in these forward-looking statements as a result of a variety of factors. While it is impossible to identify all such factors, factors that could cause actual results to differ materially from those estimated by us include the impact of general economic conditions in the United States, or in the specific markets in which we currently do business including supply chain disruptions, inflation, labor shortages and the effect on advertising activity, industry conditions, including existing competition, artificial intelligence and future competitive technologies, the popularity of radio as a broadcasting and advertising medium, cancellations, disruptions or postponements of advertising schedules in response to national or world events, our ability to develop and maintain digital technologies and hire and retain technical and sales talent, our dependence on key personnel, our capital expenditure requirements, our continued ability to identify suitable acquisition targets, and consummate and integrate any future acquisitions, legislative or regulatory requirements, risks and uncertainties relating to our leverage and changes in interest rates, our ability to obtain financing at times, in amounts and at rates considered appropriate by us, our ability to access the capital markets as and when needed and on terms that we consider favorable to us and other factors discussed in this section entitled “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in this report and under “Risk Factors” in our 2025 Annual Report on Form 10-K, for the year ended December 31, 2025, filed with the SEC on March 16, 2026, as well as other risks discussed from time to time in our filings with the SEC. Many of these factors are beyond our ability to predict or control. In addition, as a result of these and other factors, our past financial performance should not be relied on as an indication of future performance. The cautionary statements referred to in this section also should be considered in connection with any subsequent written or oral forward-looking statements that may be issued by us or persons acting on our behalf. The forward-looking statements included in this report are made only as of the date hereof or as of the date specified herein. We undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
Non-GAAP Financial Measures and Definitions
In this press release, we refer to Adjusted EBITDA, Adjusted EBITDA (Excluding Political), Adjusted Net Income and Adjusted Net Income Per Share which are financial measures that have not been prepared in accordance with generally accepted accounting principles in the United States (“GAAP”).
We define Adjusted EBITDA as net income before the deduction of income taxes, interest expense, net, (gain) loss on repurchases and extinguishment of debt, transaction and business realignment costs, depreciation and amortization, stock-based compensation, impairments, net (gain) loss on sale and retirement of assets and other expense (income), net. We define Adjusted EBITDA (Excluding Political) as Adjusted EBITDA less political net revenue, net of a fifteen percent deduction to account for estimated national representative firm fees, music licensing fees and sales commissions expense. Adjusted Net Income is defined as net income before the deduction of transaction and business realignment costs, impairments, gain on sale of investment, change in fair value of investment, net (gain) loss on sale and retirement of assets, (gain) loss on repayments, repurchases and extinguishment of debt, gain on insurance recoveries and net income attributable to non-controlling interest, net of income taxes stated at the Company’s applicable statutory effective tax rate. Adjusted Net Income Per Share is defined as Adjusted Net Income divided by the weighted average shares outstanding. We define Net Leverage as our total outstanding indebtedness, net of our total cash balance as of December 31, 2025, divided by our Adjusted EBITDA for the twelve months ended December 31, 2025. These measures do not represent, and should not be considered as alternatives to or superior to, financial results and measures determined or calculated in accordance with GAAP. In addition, these non-GAAP measures are not based on any comprehensive set of accounting rules or principles. You should be aware that in the future we may incur expenses or charges that are the same as or similar to some of the adjustments in the presentation, and we do not infer that our future results will be unaffected by unusual or nonrecurring items. In addition, these non-GAAP measures may not be comparable to similarly-named measures reported by other companies.
We use Adjusted EBITDA and Adjusted EBITDA (Excluding Political) to facilitate company-to-company operating performance comparisons by backing out potential differences caused by variations in capital structures (affecting interest expense), taxation and the age and book depreciation of facilities and equipment (affecting relative depreciation expense), which may vary for different companies for reasons unrelated to operating performance, and to facilitate year over year comparisons, by backing out the impact of political revenue which varies depending on the election cycle and may be unrelated to operating performance. We use Adjusted Net Income and Adjusted Net Income Per Share to assess total company operating performance on a consistent basis. We use Net Leverage to measure the Company’s ability to handle its debt burden. We believe that these measures, when considered together with our GAAP financial results, provide management and investors with a more complete understanding of our business operating results, including underlying trends, by excluding the effects of loss (gain) on extinguishment and repurchases of debt, transaction costs, net (gain) loss on sale and retirement of assets and investments, business realignment costs and impairments. Further, while discretionary bonuses for members of management are not determined with reference to specific targets, our board of directors may consider Adjusted EBITDA, Adjusted EBITDA (Excluding Political), Adjusted Net Income, Adjusted Net Income Per Share, and Net Leverage when determining discretionary bonuses.
Investor Relations
Claire Yenicay
(203) 900-5555
[email protected]
https://www.globenewswire.com/NewsRoom/AttachmentNg/92ff76d4-4d04-4de6-809b-09dcda7d55d7

Source: Townsquare Media Inc.