Research News and Market Data on SNAL
May 10, 2023 at 4:02 PM EDT
CULVER CITY, Calif., May 10, 2023 (GLOBE NEWSWIRE) — Snail, Inc. (Nasdaq: SNAL) (“Snail” or “the Company”), a leading, global independent developer and publisher of interactive digital entertainment, today announced financial results for the three months ended March 31, 2023.
Jim Tsai, Chief Executive Officer of Snail, commented: “We are thrilled by the ongoing engagement surrounding our ARK series. We have faced the challenging choice of postponing the release of ARK 2 until next year to ensure that we devote ample time to enhancing the game’s quality and providing an unparalleled gaming experience for our players.”
Tsai continued, “We have exciting plans to launch an expanded edition of the ARK series, which will undergo a remastering process using Unreal Engine 5. This remastered series will offer an extraordinary experience to both new and existing players, while also establishing a solid foundation for a successful launch of ARK 2.”
First Quarter 2023 and Subsequent Financial and Business Highlights
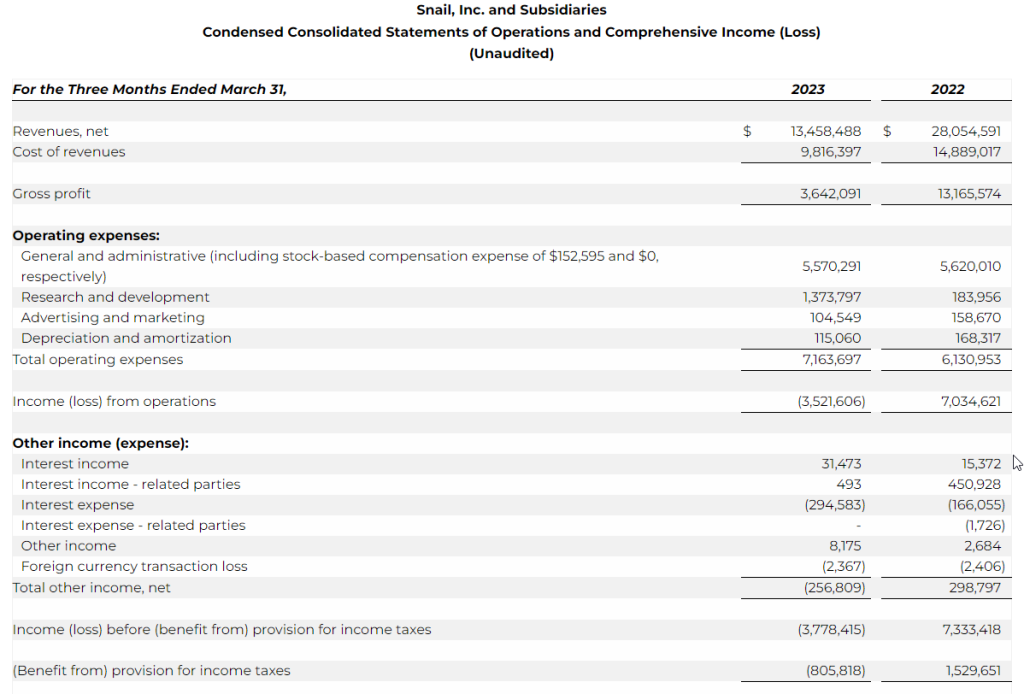
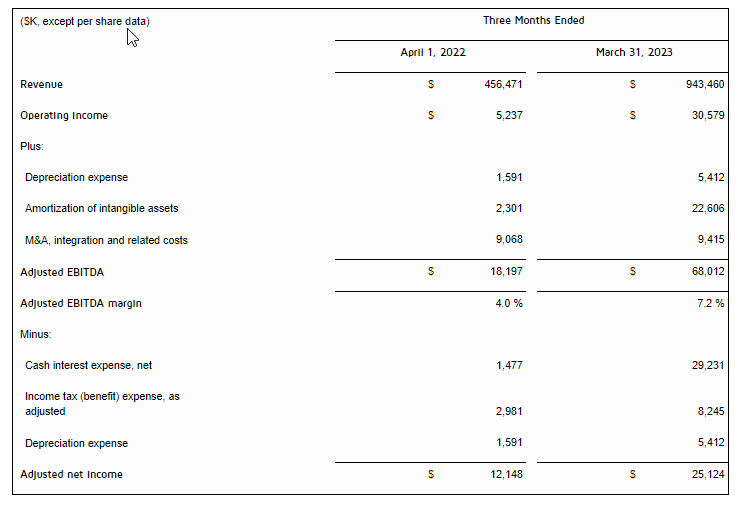
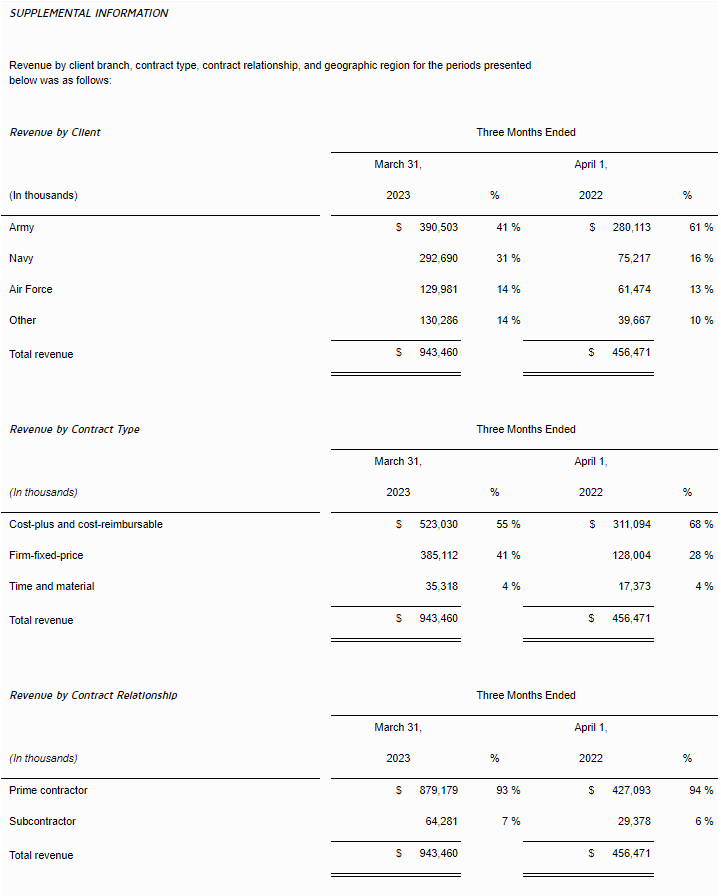
- Revenue was $13.5 million for the three months ended March 31, 2023, compared to revenue of $28.1 million in the prior year period, representing a decrease of $14.6 million. The decrease in net revenues was due to a decrease in sales of ARK, attributable to a decrease in the average sales price per unit, and the recognition of additional revenue from deferred revenue and one-time payments related to contracts with certain platforms that did not repeat in the three months ended March 31, 2023. ARK sales decreased by $3.1 million, deferred revenue from contracts decreased by $2.5 million, and one-off contract payments decreased by $8.5 million. Sales of the Company’s smaller titles decreased by a collective $0.7 million. These decreases in the Company’s smaller titles were partially offset by $0.2 million in revenue related to West Hunt.
- ARK: Survival Evolved. In the three months ended March 31, 2023, ARK: Survival Evolved averaged a total of 276,144 daily active users (“DAUs”) versus 257,168 DAUs in the prior year period.
- ARK units sold increased for the first quarter 2023 compared to the same period last year; approximately 1.6 million vs. 1.2 million, respectively.
- Through March 31, 2023, total playtime for the ARK franchise amounted to 3.2 billion hours.
- The Company sold an additional 0.4 million units of its ARK franchise in the three-month period ended March 31, 2023, versus the prior year period, due to the increase in sales promotions offered by our platform partners during the period.
- The Company expects to release ARK: Survival Ascended later this year. ARK: Survival Ascended is the entire base game of ARK: Survival Evolved, remastered with Unreal Engine 5 and expanded numerous times. It’ll feature The Island, Survival of the Fittest, and a collection of downloadable content (“DLC”) maps released over time.
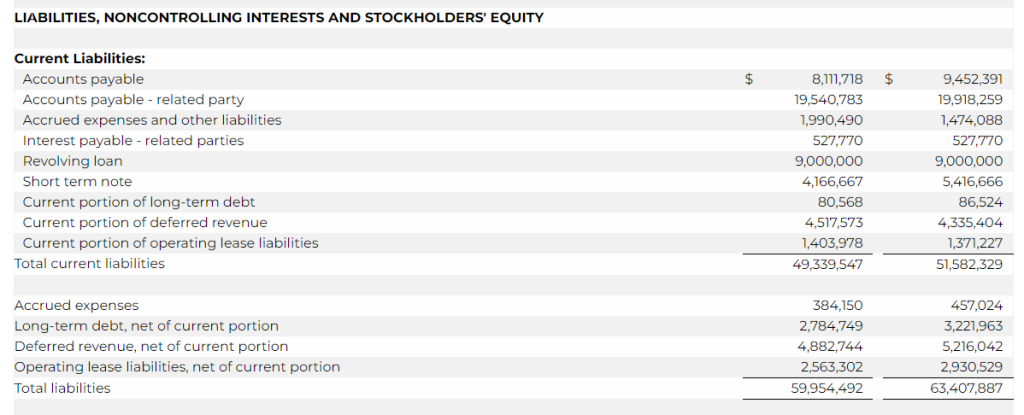
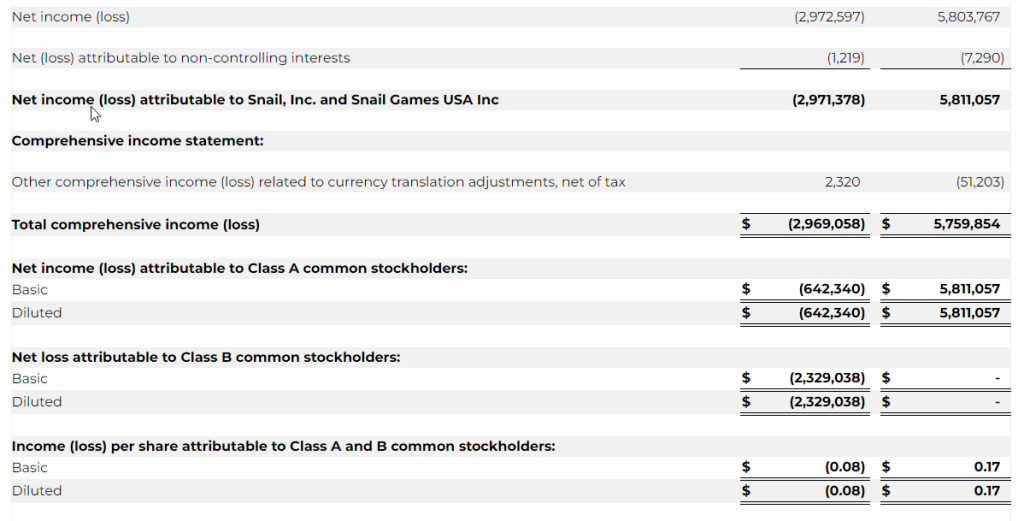
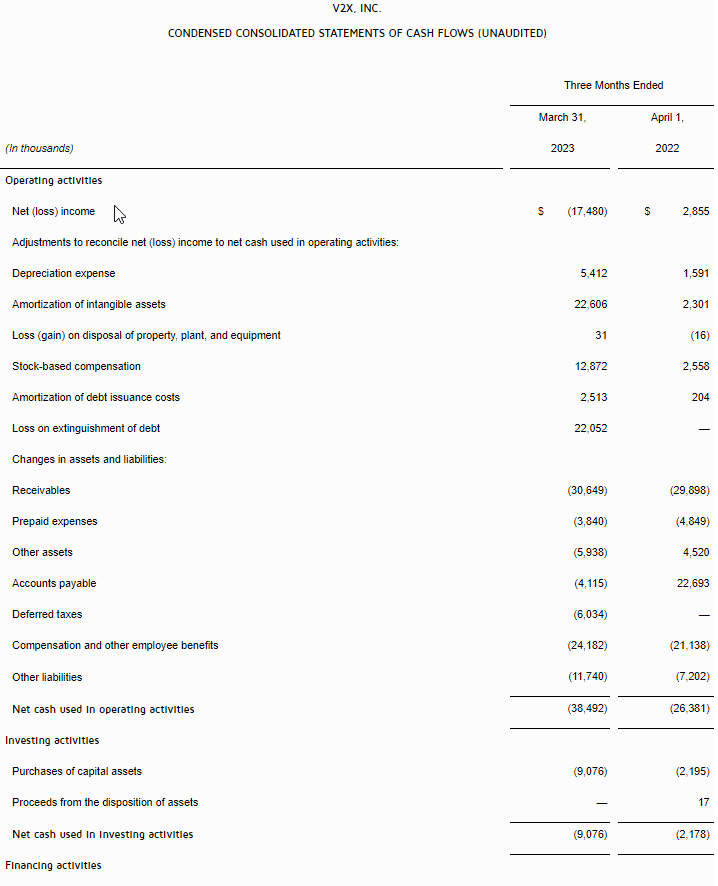
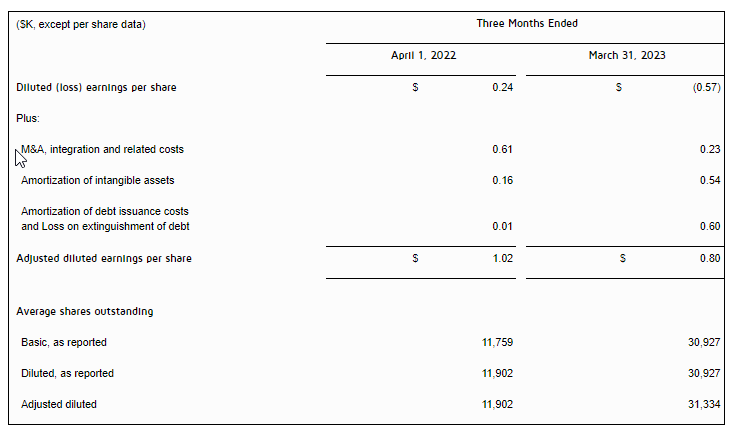
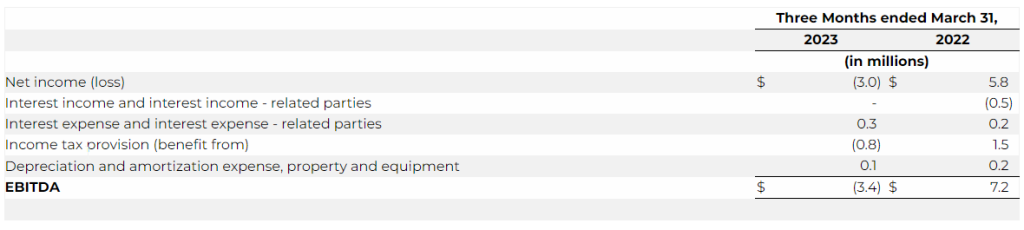
- Net loss was $3.0 million for the three months ended March 31, 2023 as compared to a net income of $5.8 million for the three months ended March 31, 2022, representing a decrease of $8.8 million. The decrease was primarily due to a decrease in revenue of $14.6 million, an increase in research and development expense of $1.2 million, a net decrease in interest income – related parties of $0.5 million, an increase in interest expense of $0.1 million, offset by a decrease in royalties of $3.2 million, a decrease in license cost and license right amortization of $1.3 million, a decrease in merchant and engine fees of $0.5 million, and a decrease in the Company’s tax provision of $2.3 million.
- Bookings for the three months ended March 31, 2023 were $13.3 million, a decrease of $12.2 million, or 47.6%, compared to the three months ended March 31, 2022. The decrease was primarily the result of decreased ARK revenues in 2023 due to the factors mentioned above.
- Earnings before interest, taxes, depreciation and amortization (“EBITDA”) for the first quarter of 2023 was a loss of $3.4 million compared to a gain of $7.2 million in the prior year period.
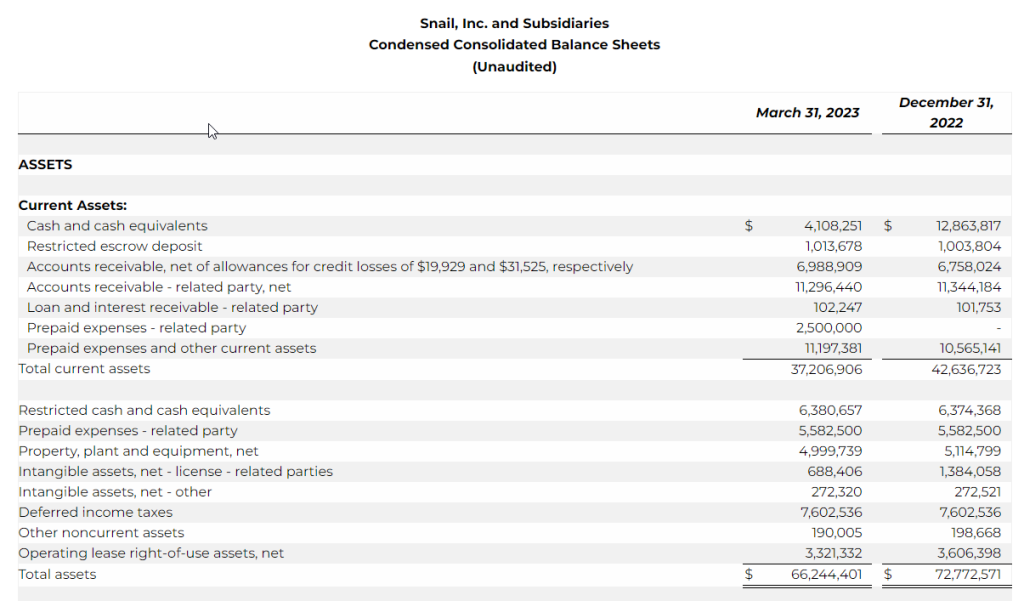
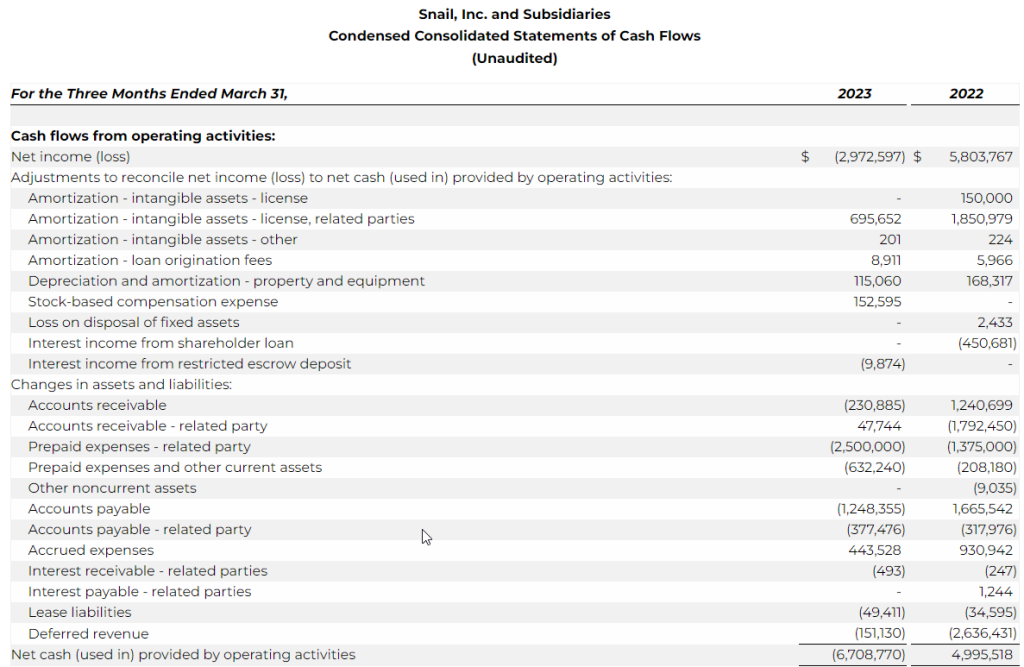
- As of March 31, 2023, unrestricted cash was $4.1 million versus $12.9 million as of December 31, 2022.
Use of Non-GAAP Financial Measures
In addition to the financial results determined in accordance with U.S. generally accepted accounting principles, or GAAP, Snail believes Bookings and EBITDA, as non-GAAP measures, are useful in evaluating its operating performance. Bookings and EBITDA are non-GAAP financial measures that are presented as supplemental disclosures and should not be construed as alternatives to net income (loss) or revenue as indicators of operating performance, nor as alternatives to cash flow provided by operating activities as measures of liquidity, both as determined in accordance with GAAP. Snail supplementally presents Bookings and EBITDA because they are key operating measures used by management to assess financial performance. Bookings adjusts for the impact of deferrals and, Snail believes, provides a useful indicator of sales in a given period. EBITDA adjusts for items that Snail believes do not reflect the ongoing operating performance of its business, such as certain non-cash items, unusual or infrequent items or items that change from period to period without any material relevance to its operating performance. Management believes Bookings and EBITDA are useful to investors and analysts in highlighting trends in Snail’s operating performance, while other measures can differ significantly depending on long-term strategic decisions regarding capital structure, the tax jurisdictions in which Snail operates and capital investments.
Bookings is defined as the net amount of products and services sold digitally or physically in the period. Bookings is equal to revenues excluding the impact from deferrals. Below is a reconciliation of total net revenue to Bookings, the closest GAAP financial measure.

We define EBITDA as net income (loss) before (i) interest expense, (ii) interest income, (iii) income tax provision (benefit from) and (iv) depreciation and amortization expense. The following table provides a reconciliation from net income (loss) to EBITDA:
Webcast Details
The Company will host a webcast at 5:00 PM ET today to discuss the first quarter 2023 financial results. Participants may access the live webcast and replay on the Company’s investor relations website at https://investor.snail.com/. The earnings call may also be accessed by dialling 1 (877) 451-6152 from the United States, or by dialling 1 (201) 389-0879 internationally.
About Snail, Inc.
Snail is a leading, global independent developer and publisher of interactive digital entertainment for consumers around the world, with a premier portfolio of premium games designed for use on a variety of platforms, including consoles, PCs and mobile devices.
Contacts:
Investors:
[email protected]
Forward-Looking Statements
This press release contains statements that constitute forward-looking statements. Many of the forward-looking statements contained in this press release can be identified by the use of forward-looking words such as “anticipate,” “believe,” “could,” “expect,” “should,” “plan,” “intend,” “may,” “predict,” “continue,” “estimate” and “potential,” or the negative of these terms or other similar expressions. Forward-looking statements appear in a number of places in this press release and include, but are not limited to, statements regarding Snail’s intent, belief or current expectations. These forward-looking statements include information about possible or assumed future results of Snail’s business, financial condition, results of operations, liquidity, plans and objectives. The statements Snail makes regarding the following matters are forward-looking by their nature: growth prospects and strategies; launching new games and additional functionality to games that are commercially successful; expectations regarding significant drivers of future growth; its ability to retain and increase its player base and develop new video games and enhance existing games; competition from companies in a number of industries, including other casual game developers and publishers and both large and small, public and private Internet companies; its ability to attract and retain a qualified management team and other team members while controlling its labor costs; its relationships with third-party platforms such as Xbox Live and Game Pass, PlayStation Network, Steam, Epic Games Store, My Nintendo Store, the Apple App Store, the Google Play Store and the Amazon Appstore; the size of addressable markets, market share and market trends; its ability to successfully enter new markets and manage international expansion; protecting and developing its brand and intellectual property portfolio; costs associated with defending intellectual property infringement and other claims; future business development, results of operations and financial condition; the ongoing conflict involving Russia and Ukraine on its business and the global economy generally; rulings by courts or other governmental authorities; the Share Repurchase Program, including expectations regarding the timing and manner of repurchases made under the program; its plans to pursue and successfully integrate strategic acquisitions; assumptions underlying any of the foregoing.
Further information on risks, uncertainties and other factors that could affect Snail’s financial results are included in its filings with the Securities and Exchange Commission (the “SEC”) from time to time, annual reports on Forms 10-K and quarterly reports on 10-Q filed, or to be filed, with the SEC. You should not rely on these forward-looking statements, as actual outcomes and results may differ materially from those expressed or implied in the forward-looking statements as a result of such risks and uncertainties. All forward-looking statements in this press release are based on management’s beliefs and assumptions and on information currently available to Snail, and Snail does not assume any obligation to update the forward-looking statements provided to reflect events that occur or circumstances that exist after the date on which they were made.