Research News and Market Data on KTOS
August 7, 2025 at 4:00 PM EDT
Second Quarter 2025 Revenues of $351.5 Million Reflect 17.1 Percent Growth and 15.2 Percent Organic Growth Over Second Quarter 2024 Revenues of $300.1 Million
Second Quarter 2025 Consolidated Book to Bill Ratio of 0.7 to 1 and Bookings of $257.0 Million
Last Twelve Months Ended June 29, 2025 Consolidated Book to Bill Ratio of 1.2 to 1 and Bookings of $1.401 Billion
Increases 2025 Full Year Revenue and Adjusted EBITDA Guidance
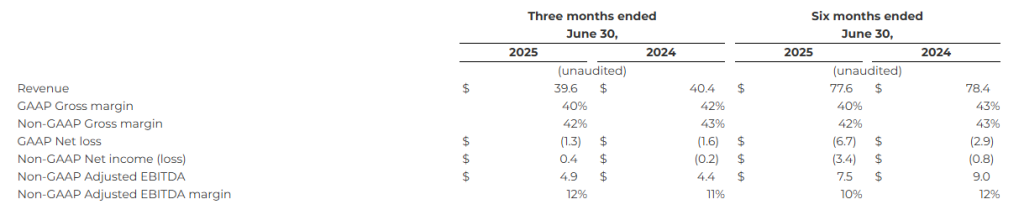
SAN DIEGO, Aug. 07, 2025 (GLOBE NEWSWIRE) — Kratos Defense & Security Solutions, Inc. (Nasdaq: KTOS), a Technology Company in the Defense, National Security and Commercial Markets, today reported its second quarter 2025 financial results, including Revenues of $351.5 million, Operating Income of $3.7 million, Net Income of $2.9 million, Adjusted EBITDA of $28.3 million and a consolidated book to bill ratio of 0.7 to 1.0.
Second quarter 2025 Net Income and Operating Income includes non-cash stock compensation expense of $8.6 million, and Company-funded Research and Development (R&D) expense of $10.2 million, including efforts in our Space, Satellite, Unmanned Systems and Microwave Electronic businesses, and expense to accrue $2.0 million related to settlement of a legal matter, offset partially by non-recurring credits of $1.1 million related to the resolution of a previously recorded contingent liability.
Kratos reported second quarter 2025 GAAP Net Income of $2.9 million and GAAP Net Income per share of $0.02, compared to GAAP Net Income of $7.9 million and GAAP Net Income per share of $0.05, for the second quarter of 2024. Adjusted earnings per share (EPS) was $0.11 for the second quarter of 2025, compared to $0.14 for the second quarter of 2024.
Second quarter 2025 Revenues of $351.5 million increased $51.4 million, reflecting 15.2 percent organic growth from second quarter 2024 Revenues of $300.1 million. Organic revenue growth was reported in our KGS segment of 27.1 percent, with the most notable growth in our Defense Rocket Systems and C5ISR businesses, with organic revenue growth rates of 116.6 percent and 25.4 percent, respectively, compared to the second quarter of 2024.
Second quarter 2025 Cash Flow Used in Operations was $10.6 million, primarily reflecting the working capital requirements related to the revenue growth impacting our receivables, increases in inventories for anticipated future deliveries and ramps in production and investments we are making related to certain development initiatives in our Unmanned Systems (KUS) segment. Free Cash Flow Used in Operations for the second quarter of 2025 was $31.1 million after funding of $20.5 million of capital expenditures.
For the second quarter of 2025, KUS generated Revenues of $73.2 million, compared to $85.8 million in the second quarter of 2024, which included revenues of $17.4 million related to an international target drone shipment made during the second quarter of 2024. KUS’s Operating Loss was $0.3 million in the second quarter of 2025, compared to Operating Income of $3.6 million in the second quarter of 2024. KUS’s Adjusted EBITDA for the second quarter of 2025 was $3.6 million, compared to $7.2 million for the second quarter of 2024, reflecting the impact of the revenue volume and continued impact of increased material and subcontractor and labor costs on multi-year fixed price production contracts under terms which were negotiated in 2020 and 2021 that we are unable to seek recovery and which we are unable to renegotiate until the next multi-year production lot.
KUS’s book-to-bill ratio for the second quarter of 2025 was 0.9 to 1.0 and 1.3 to 1.0 for the twelve months ended June 29, 2025, with bookings of $63.7 million for the three months ended June 29, 2025, and bookings of $359.5 million for the twelve months ended June 29, 2025. Total backlog for KUS at the end of the second quarter of 2025 was $337.6 million, compared to $347.1 million at the end of the first quarter of 2025.
For the second quarter of 2025, Kratos’ Government Solutions (KGS) segment Revenues of $278.3 million increased from Revenues of $214.3 million in the second quarter of 2024, reflecting a 27.1 percent organic growth rate, excluding the impact of the recent acquisition of certain assets of Norden Millimeter, Inc. The increased Revenues includes organic revenue growth across all KGS businesses, with the most notable growth in our Defense Rocket Support business which includes the timing of certain hypersonic missions, and in our C5ISR businesses with organic revenue growth rates of 116.6 percent and 25.4 percent, respectively, over the second quarter of 2024.
KGS reported Operating Income of $12.6 million in the second quarter of 2025 compared to $15.5 million in the second quarter of 2024, primarily reflecting the mix in revenues, particularly impacted by a less favorable mix in our Space, Training and Cyber business. Second quarter 2025 KGS Adjusted EBITDA was $24.7 million, compared to second quarter 2024 KGS Adjusted EBITDA of $22.7 million, primarily reflecting the mix in revenues and resources.
KGS reported a book-to-bill ratio of 0.7 to 1.0 for the second quarter of 2025, a book to bill ratio of 1.2 to 1.0 for the last twelve months ended June 29, 2025 and bookings of $193.3 million and $1.041 billion for the three and last twelve months ended June 29, 2025, respectively. KGS’s total backlog was $1.076 billion at the end of the second quarter of 2025, compared to $1.161 billion at the end of the first quarter of 2025.
Kratos reported consolidated bookings of $257.0 million and a book-to-bill ratio of 0.7 to 1.0 for the second quarter of 2025, and consolidated bookings of $1.401 billion and a book-to-bill ratio of 1.2 to 1.0 for the last twelve months ended June 29, 2025. Consolidated backlog was $1.414 billion on June 29, 2025, as compared to $1.508 billion on March 30, 2025. Kratos’ bid and proposal pipeline was $13.0 billion at June 29, 2025, as compared to $12.6 billion at March 30, 2025. Backlog at June 29, 2025, included funded backlog of $1.125 billion and unfunded backlog of $288.6 million.
Eric DeMarco, Kratos’ President and CEO, said, “A generational recapitalization of strategic weapon systems is underway, with significant global funding being committed by the U.S. and its allies, including as represented by a planned U.S. 2026 National Security spend exceeding $1 trillion, NATO committing 5% of member GDP to defense, and Asian allies looking to do the same. The Global Defense and National Security Market is currently approximately $2.5 trillion, led by the United States “peace through strength” posture and is expected to grow for the foreseeable future. Since our last report to you, President Trump signed the Reconciliation Bill into law, supplementing defense spending by an additional $150 billion, including increased funding for drones, air defense systems, missiles, radars, space and satellites and the Golden Dome initiative, each related to primary Kratos capability areas.”
Mr. DeMarco continued, “In addition to significant increased funding expected for our industry, Secretary of Defense Pete Hegseth recently announced sweeping changes to the way the Pentagon will buy and field unmanned aerial systems, which is related to a June 6, 2025 Presidential Executive order on “Unleashing American Drone Dominance”, both expected to accelerate the acquisition and fielding of drones. Also, further emphasizing the new focus on streamlining defense acquisition, promoting innovation and commercial “ready to go” solutions to deliver capabilities to the warfighter faster and more efficiently, Senator Roger Wicker recently introduced the FORGED Act and House Armed Services Committee Chair Mike Rogers and Ranking Member Adam Smith introduced the related SPEED Act, all of which, if enacted, we expect to benefit Kratos and our “First to Market” strategy.”
Mr. DeMarco added, “We believe that we are seeing direct positive impact from these actions, including Kratos’ Q225 17 percent revenue growth rate, our near record backlog, our record $13 billion opportunity pipeline and LTM book–to–bill ratio of 1.2 to 1.0. Additionally, since the end of the second quarter, we have been informed that we have been successful as prime, on a new, single award, potential total value up to $750 million program, internal code name Poseidon, which we expect to receive a formal contract by the end of this year. Additionally, we were also recently informed that Kratos, with our strategic partner, were down selected on another large, new program of record opportunity we call Deimos”.
Mr. DeMarco went on, “Virtually every Kratos business unit is forecasting significant future organic growth, including our hypersonic system franchise, small jet engines for drones, missiles and loitering munitions, our Israeli based microwave electronics business, and our military grade hardware business supporting missile, radar, hypersonic, counter UAS and strategic weapon systems. We are also expecting increased EBITDA margins beginning in 2026 and continuing thereafter, as new higher margin programs begin, certain lower margin contracts are renegotiated at renewal, and as we reduce costs in certain areas. We expect to continue to make important investments in property, plant, facilities, equipment etc., over the next two years, in conjunction with our funded customers, partners, initiatives and programs, to capture new opportunities, rebuild the industrial base, further grow our business and generate additional value for all Kratos stakeholders.”
Mr. DeMarco concluded, “In 2015, Kratos Mako tactical jet drones flew manned-unmanned teaming with Marine Corps Harrier jet aircraft, establishing Kratos as a first-to-market innovation leader, delivering actual relevant products, not PowerPoints, to the customer. As Kratos invests its own money in research and product development, we are incentivized to move fast, do it right, at a low cost, and always with an eye on being able to produce our military grade products, hardware and software affordably, at scale, and generate an adequate return for our shareholders. We believe that Kratos is in the right places, at the right time, with the right products, and importantly at the right affordable price points, to address our customer and partners requirements, to deliver significant value to Kratos stakeholders today.”
Financial Guidance
We are providing our initial 2025 third quarter guidance and increasing our full year 2025 Revenue and Adjusted EBITDA guidance range, which includes our assumptions, including as related to: current forecasted business mix, employee sourcing, hiring and retention; manufacturing, production and supply chain disruptions; parts shortages and related continued significant cost and price increases in each of these areas, that are impacting the industry and Kratos.
Kratos’ 2025 financial forecast and guidance includes elevated investments for capital expenditures for property, plant and equipment, including the expansion of our manufacturing and production facilities and related inventory builds in our Rocket Systems and Hypersonic businesses, primarily related to the recent MACH-TB 2.0 contract award, the continued manufacture of two production lots of Valkyries prior to contract award, to meet anticipated customer orders and requirements, the expansion and build-out of the Company’s Microwave Products production facilities, the expansion and build-out of our small jet engine production and test cell facilities, and the build-out of additional secure facilities for our federal secured space communications business, in accordance with contract and customer requirements. Kratos’ operating cash flow guidance also assumes consummation of certain investments in our rocket systems and unmanned systems businesses.
Our third quarter and full year 2025 guidance ranges are as follows:
| Current Guidance Range | ||
| $M | Q325 | FY25 |
| Revenues | $315 – $325 | $1,290 – $1,310 |
| R&D | $10 – $11 | $40 – $42 |
| Operating Income | $3 – $7 | $29 – $34 |
| Depreciation | $9 – $10 | $35 – $38 |
| Amortization | $3 – $4 | $12 – $14 |
| Stock Based Compensation | $8 – $9 | $34 – $36 |
| Adjusted EBITDA | $25 – $30 | $114 – $120 |
| Operating Cash Flow | $50 – $60 | |
| Capital Expenditures | $125 – $135 | |
| Free Cash Flow Use | ($75 – $85) | |
Management will discuss the Company’s financial results on a conference call beginning at 2:00 p.m. Pacific (5:00 p.m. Eastern) today. The call will be available at www.kratosdefense.com. Participants may register for the call using this Online Form. Upon registration, all telephone participants will receive the dial-in number along with a unique PIN that can be used to access the call. For those who cannot access the live broadcast, a replay will be available on Kratos’ website.
About Kratos Defense & Security Solutions
Kratos Defense & Security Solutions, Inc. (NASDAQ: KTOS) is a technology, products, system and software company addressing the defense, national security, and commercial markets. Kratos makes true internally funded research, development, capital and other investments, to rapidly develop, produce and field solutions that address our customers’ mission critical needs and requirements. At Kratos, affordability is a technology, and we seek to utilize proven, leading edge approaches and technology, not unproven bleeding edge approaches or technology, with Kratos’ approach designed to reduce cost, schedule and risk, enabling us to be first to market with cost effective solutions. We believe that Kratos is known as an innovative disruptive change agent in the industry, a company that is an expert in designing products and systems up front for successful rapid, large quantity, low-cost future manufacturing which is a value add competitive differentiator for our large traditional prime system integrator partners and also to our government and commercial customers. Kratos intends to pursue program and contract opportunities as the prime or lead contractor when we believe that our probability of win (PWin) is high and any investment required by Kratos is within our capital resource comfort level. We intend to partner and team with a large, traditional system integrator when our assessment of PWin is greater or required investment is beyond Kratos’ comfort level. Kratos’ primary business areas include virtualized ground systems for satellites and space vehicles including software for command & control (C2) and telemetry, tracking and control (TT&C), jet powered unmanned aerial drone systems, hypersonic vehicles and rocket systems, propulsion systems for drones, missiles, loitering munitions, supersonic systems, space craft and launch systems, C5ISR and microwave electronic products for missile, radar, missile defense, space, satellite, counter UAS, directed energy, communication and other systems, and virtual & augmented reality training systems for the warfighter. For more information, visit www.KratosDefense.com.
Notice Regarding Forward–Looking Statements
This news release contains certain forward-looking statements that involve risks and uncertainties, including, without limitation, express or implied statements concerning the Company’s expectations regarding its future financial performance, including the Company’s expectations for its third quarter and full year 2025 revenues, R&D, operating income, depreciation, amortization, stock based compensation expense, and Adjusted EBITDA, and full year 2025 operating cash flow, capital expenditures, and free cash flow, forecasted business unit organic revenue growth, EBITDA margins in 2026 and thereafter, future initiation of higher margin programs and negotiation of lower margin contracts which are expected to be renewed in the future, expected future investments in property, plant, facilities, and equipment, the Company’s bid and proposal pipeline and backlog, including the Company’s ability to timely execute on its backlog, demand for its products and services, including the Company’s alignment with today’s National Security requirements and the positioning of its C5ISR and other businesses, ability to successfully compete and expected new customer awards, the impact of the Company’s restructuring efforts and cost reduction measures, the availability and timing of government funding for the Company’s offerings, availability of an experienced skilled workforce, inflation and increased costs, risks related to potential cybersecurity events or disruptions of our information technology systems, and delays in our financial projections, industry, business and operations, including projected growth. Such statements are only predictions, and the Company’s actual results may differ materially from the results expressed or implied by these statements. Investors are cautioned not to place undue reliance on any such forward-looking statements. All such forward-looking statements speak only as of the date they are made, and the Company undertakes no obligation to update or revise these statements, whether as a result of new information, future events or otherwise. Factors that may cause the Company’s results to differ include, but are not limited to: risks to our business and financial results related to the reductions and other spending constraints imposed on the U.S. Government and our other customers, including as a result of sequestration and extended continuing resolutions, the Federal budget deficit and Federal government shut-downs; risks of adverse regulatory action or litigation; risks associated with debt leverage; risks that our cost-cutting initiatives will not provide the anticipated benefits; risks that changes, cutbacks or delays in spending by the DoD may occur, which could cause delays or cancellations of key government contracts; risks of delays to or the cancellation of our projects as a result of protest actions submitted by our competitors; risks that changes may occur in Federal government (or other applicable) procurement laws, regulations, policies and budgets; risks of the availability of government funding for the Company’s products and services due to performance, cost growth, or other factors, changes in government and customer priorities and requirements (including cost-cutting initiatives, the potential deferral of awards, terminations or reduction of expenditures to respond to the priorities of Congress and the Administration, or budgetary cuts resulting from Congressional committee recommendations or automatic sequestration under the Budget Control Act of 2011, as amended); risks that the unmanned aerial systems and unmanned ground sensor markets do not experience significant growth; risks that products we have developed or will develop will not become programs of record; risks that we cannot expand our customer base or that our products do not achieve broad acceptance which could impact our ability to achieve our anticipated level of growth; risks of increases in the Federal government initiatives related to in-sourcing; risks related to security breaches, including cyber security attacks and threats or other significant disruptions of our information systems, facilities and infrastructures; risks related to our compliance with applicable contracting and procurement laws, regulations and standards; risks related to the new DoD Cybersecurity Maturity Model Certification; risks relating to the ongoing conflict in Ukraine and the Israeli-Palestinian military conflict; risks to our business in Israel; risks related to contract performance; risks related to failure of our products or services; risks associated with our subcontractors’ or suppliers’ failure to perform their contractual obligations, including the appearance of counterfeit or corrupt parts in our products; changes in the competitive environment (including as a result of bid protests); failure to successfully integrate acquired operations and compete in the marketplace, which could reduce revenues and profit margins; risks that potential future goodwill impairments will adversely affect our operating results; risks that anticipated tax benefits will not be realized in accordance with our expectations; risks that a change in ownership of our stock could cause further limitation to the future utilization of our net operating losses; risks that we may be required to record valuation allowances on our net operating losses which could adversely impact our profitability and financial condition; risks that the current economic environment will adversely impact our business, including with respect to our ability to recruit and retain sufficient numbers of qualified personnel to execute on our programs and contracts, as well as expected contract awards and risks related to increasing interest rates and risks related to the interest rate swap contract to hedge Term SOFR associated with the Company’s Term Loan A; currently unforeseen risks associated with any public health crisis, and risks related to natural disasters or severe weather. These and other risk factors are more fully discussed in the Company’s Annual Report on Form 10-K for the period ended December 29, 2024, and in our other filings made with the Securities and Exchange Commission.
Note Regarding Use of Non-GAAP Financial Measures and Other Performance Metrics
This news release contains non-GAAP financial measures, including organic revenue growth rates, Adjusted EPS (computed using income before income taxes, excluding depreciation, amortization of intangible assets, amortization of capitalized contract and development costs, stock-based compensation expense, acquisition and restructuring related items and other, which includes, but is not limited to, legal related items, non-recoverable rates and costs, and foreign transaction gains and losses, less the estimated impact to income taxes) and Adjusted EBITDA (which excludes, among other things, acquisition and restructuring related items, stock compensation expense, foreign transaction gains and losses, and the associated margin rates). Additional non-GAAP financial measures include Free Cash Flow from Operations computed as Cash Flow from Operations less Capital Expenditures plus proceeds from sale of assets and Adjusted EBITDA related to our KUS and KGS businesses. Kratos believes this information is useful to investors because it provides a basis for measuring the Company’s available capital resources, the actual and forecasted operating performance of the Company’s business and the Company’s cash flow, excluding non-recurring items and non-cash items that would normally be included in the most directly comparable measures calculated and presented in accordance with GAAP. The Company’s management uses these non-GAAP financial measures, along with the most directly comparable GAAP financial measures, in evaluating the Company’s actual and forecasted operating performance, capital resources and cash flow. Non-GAAP financial measures should not be considered in isolation from, or as a substitute for, financial information presented in compliance with GAAP, and investors should carefully evaluate the Company’s financial results calculated in accordance with GAAP and reconciliations to those financial results. In addition, non-GAAP financial measures as reported by the Company may not be comparable to similarly titled amounts reported by other companies. As appropriate, the most directly comparable GAAP financial measures and information reconciling these non-GAAP financial measures to the Company’s financial results prepared in accordance with GAAP are included in this news release.
Another Performance Metric the Company believes is a key performance indicator in our industry is our Book to Bill Ratio as it provides investors with a measure of the amount of bookings or contract awards as compared to the amount of revenues that have been recorded during the period and provides an indicator of how much of the Company’s backlog is being burned or utilized in a certain period. The Book to Bill Ratio is computed as the number of bookings or contract awards in the period divided by the revenues recorded for the same period. The Company believes that the rolling or last twelve months’ Book to Bill Ratio is meaningful since the timing of quarter-to-quarter bookings can vary.
| Press Contact: Claire Burghoff claire.burghoff@kratosdefense.com Investor Information: 877-934-4687 investor@kratosdefense.com |

Source: Kratos Defense & Security Solutions, Inc.