Research News and Market Data on UNCY
November 13, 2024 7:15am EST Download as PDF
– OLC New Drug Application (NDA) Accepted by the FDA with a PDUFA Target Action Date of June 28, 2025–
– Commercial Planning in Progress for 2025 Launch –
– Late Breaker Poster Presentation on OLC at ASN Kidney Week –
– Successful Completion of UNI-494 Phase 1 Trial –
LOS ALTOS, Calif., Nov. 13, 2024 (GLOBE NEWSWIRE) — Unicycive Therapeutics, Inc. (Nasdaq: UNCY) (the “Company” or “Unicycive”), a clinical-stage biotechnology company developing therapies for patients with kidney disease, today announced its financial results for the three months ended September 30, 2024, and provided a business update.
“We are pleased with the tremendous progress we have made over the last several months highlighted by the acceptance of our New Drug Application for oxylanthanum carbonate (OLC) which may result in the potential approval of our first drug in 2025,” said Shalabh Gupta, M.D., Chief Executive Officer of Unicycive. “If approved, we believe OLC’s high potency and low pill burden would provide a best-in-class option for patients with chronic kidney disease (CKD) on dialysis who have hyperphosphatemia and face adherence challenges with current treatment regimens. With the NDA acceptance now behind us, we are actively preparing to commercialize OLC with the goal of bringing this innovative new treatment to market in the second half of 2025.”
“We have also made progress on our second asset, UNI-494, as we announced the successful completion of our Phase 1 clinical trial providing the necessary data to potentially advance to Phase 2 clinical development. UNI-494 is targeting acute kidney injury (AKI), a challenging and often under-treated disease. We plan to request a meeting with the FDA by the end of this year to continue advancing our clinical development program for UNI-494,” concluded Dr. Gupta.
Key Highlights
- Announced the acceptance of the New Drug Application (NDA) by the U.S. Food and Drug Administration (FDA) for OLC for the treatment of hyperphosphatemia in patients with CKD on dialysis. The FDA set a Prescription Drug User Fee Act (PDUFA) Target Action Date of June 28, 2025.
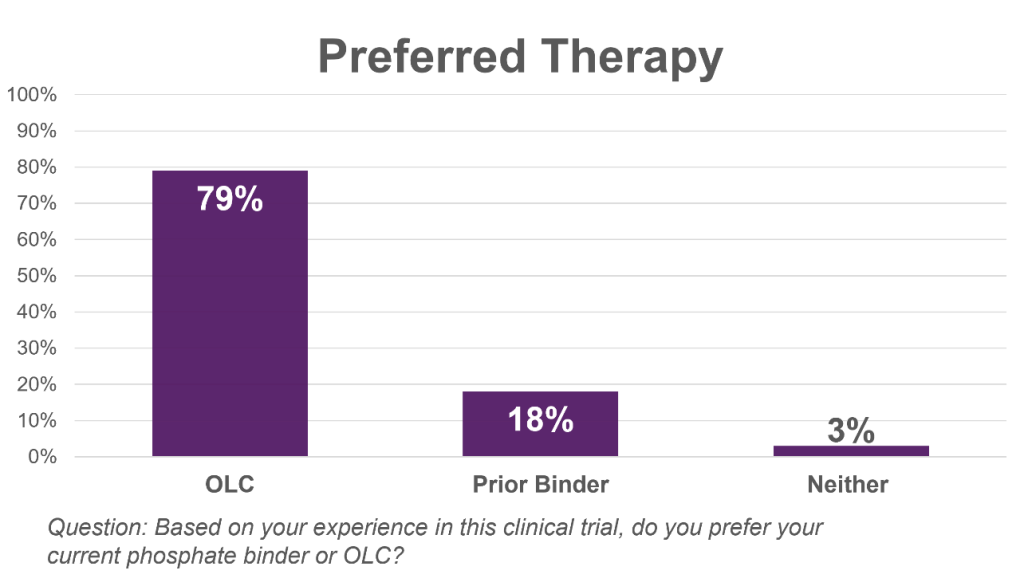
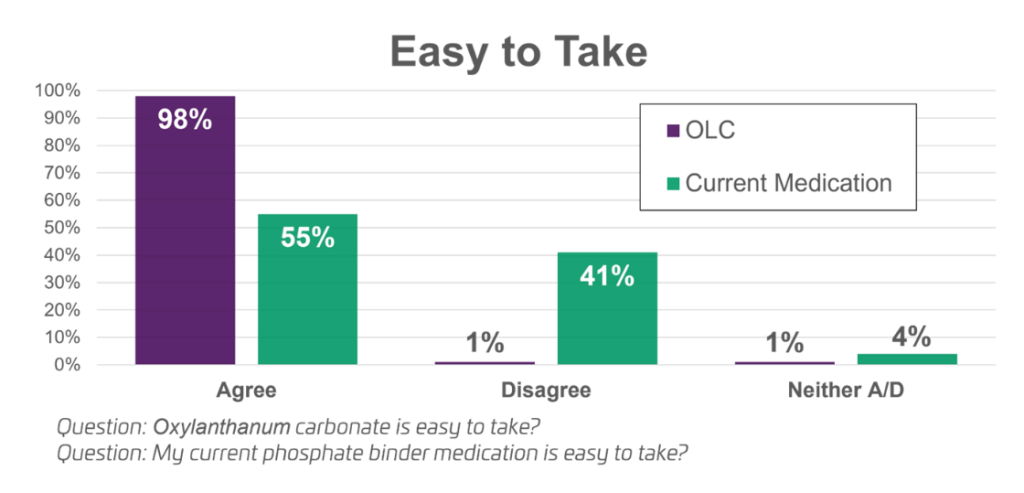
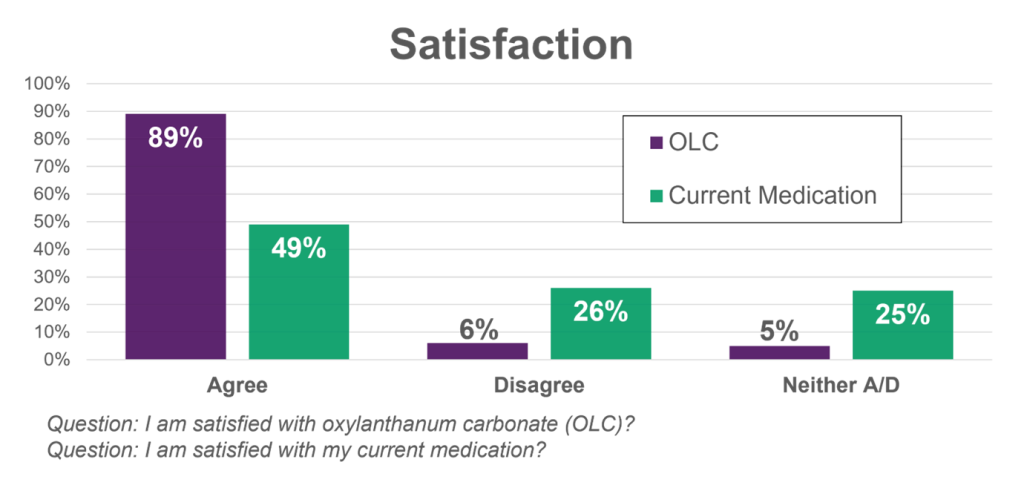
- Announced initial results from the patient reported outcome survey conducted during the UNI-OLC-201 pivotal clinical trial. In the survey, OLC consistently outperformed the other phosphate binders in all categories: 79% of patients preferred OLC compared to 18% of patients who preferred their prior therapy; 98% of patients said that OLC was easy to take compared to 55% for their prior therapy; and 89% of patients said they were satisfied with OLC while only 49% were satisfied with their prior therapy.
- Announced the successful completion of the UNI-494 Phase 1 study in healthy volunteers. The Phase 1 study was a single center, double-blind, placebo-controlled, randomized single ascending dose (Part 1) and multiple ascending dose (Part 2) study in healthy volunteers conducted in the United Kingdom. UNI-494 was well-tolerated as a single dose up to 160 mg and in multiple doses at 40 mg twice-a-day. The absorption of UNI-494 was fast, and UNI-494 was rapidly metabolized to release nicorandil. Collectively, the results will help determine the dose and schedule of UNI-494 in a potential Phase 2 clinical trial in patients with acute kidney injury.
- Granted a patent on UNI-494 to treat AKI by the United States Patent and Trademark Office (USPTO). The patent, valid until 2040, secures protection of a method of treating a disease or a condition (“method of use”) related to AKI or contrast-induced-nephropathy by administering the UNI-494 compound.
- Delivered four presentations on OLC and UNI-494 at the American Society of Nephrology (ASN) Kidney Week 2024 including a late-breaker poster presentation highlighting favorable safety and tolerability data of OLC. The presentation highlighted the positive pivotal clinical trial data demonstrating that OLC was able to achieve serum phosphate control in more than 90% of patients with CKD on dialysis who entered the maintenance phase of the trial. A poster presentation also described the results from the UNI-494 Phase 1 study results, and two preclinical posters were presented.
- Two preclinical studies for both OLC and UNI-494 were featured in two publications: “Systemic Absorption of Oxylanthanum Carbonate is Minimal in Preclinical Models” was published in the Pharmaceutical Chemistry Journal; “Evaluation of UNI-494 in Acute Kidney Injury Treatment Efficacy When Administered After Ischemia-Reperfusion in a Rat Model” was published in EC Pharmacology and Toxicology.
- UNCY was added to the Russell Microcap® Index effective July 1, 2024. Membership in the Russell Microcap® Index, which remains in place for one year is accompanied by automatic inclusion in the appropriate growth and value style indexes.
Financial Results for the Quarter Ended September 30, 2024
Research and Development (R&D) expenses were $3.0 million for the three months ended September 30, 2024, compared to $3.4 million for the three months ended September 30, 2023. The decrease in research and development expenses was primarily due to decreased drug development costs.
General and Administrative (G&A) expenses were $3.2 million for the three months ended September 30, 2024, compared to $2.6 million for the three months ended September 30, 2023. The increase was primarily due to increased non-cash stock compensation expense.
Other Income was $2.2 million for the three months ended September 30, 2024 compared to $1.6 million in the three months ended September 30, 2023, due primarily to a decrease in the fair value of our warrant liability.
Net loss attributable to common stockholders for the three months ended September 30, 2024 was $4.1 million, compared to a net loss attributable to common stockholders of $4.4 million, for the three months ended September 30, 2023. The decreased net loss for the three-month period ended September 30, 2024 was attributable to a decrease in the fair value of our warrant liability.
As of September 30, 2024, cash and cash equivalents totaled $32.3 million. The Company believes that it has sufficient resources to fund planned operations into 2026.
About Unicycive Therapeutics
Unicycive Therapeutics is a biotechnology company developing novel treatments for kidney diseases. Unicycive’s lead drug candidate, oxylanthanum carbonate (OLC), is a novel investigational phosphate binding agent being developed for the treatment of hyperphosphatemia in chronic kidney disease patients on dialysis. Positive pivotal trial results were reported in June 2024 for OLC, and a New Drug Application (NDA) is under review by the U.S. Food and Drug Administration (FDA) with a Prescription Drug User Fee Act (PDUFA) Target Action Date of June 28, 2025. OLC is protected by a strong global patent portfolio including an issued patent on composition of matter with exclusivity until 2031, and with the potential patent term extension until 2035 after OLC approval. Unicycive’s second asset, UNI-494, is a patent-protected new chemical entity in clinical development for the treatment of conditions related to acute kidney injury. UNI-494 has successfully completed a Phase 1 trial. For more information, please visit Unicycive.com and follow us on LinkedIn and YouTube.
Forward-looking statements
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. These statements may be identified using words such as “anticipate,” “believe,” “forecast,” “estimated” and “intend” or other similar terms or expressions that concern Unicycive’s expectations, strategy, plans or intentions. These forward-looking statements are based on Unicycive’s current expectations and actual results could differ materially. There are several factors that could cause actual events to differ materially from those indicated by such forward-looking statements. These factors include, but are not limited to, clinical trials involve a lengthy and expensive process with an uncertain outcome, and results of earlier studies and trials may not be predictive of future trial results; our clinical trials may be suspended or discontinued due to unexpected side effects or other safety risks that could preclude approval of our product candidates; risks related to business interruptions which could seriously harm our financial condition and increase our costs and expenses; dependence on key personnel; substantial competition; uncertainties of patent protection and litigation; dependence upon third parties; and risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the uncertainties related to market conditions and other factors described more fully in the section entitled ‘Risk Factors’ in Unicycive’s Annual Report on Form 10-K for the year ended December 31, 2023, and other periodic reports filed with the Securities and Exchange Commission. Any forward-looking statements contained in this press release speak only as of the date hereof, and Unicycive specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events or otherwise.
Investor Contact:
Kevin Gardner
LifeSci Advisors
kgardner@lifesciadvisors.com
Chris Calabrese
LifeSci Advisors
ccalabrese@lifesciadvisors.com
SOURCE: Unicycive Therapeutics, Inc.