Research News and Market Data on ELDN
March 20, 2025
PDF Version
Tegoprubart used as a key component of immunosuppression regimen in the second transplant of a genetically modified pig kidney into a human conducted at Massachusetts General Hospital
Announced positive initial data from first three subjects with type 1 diabetes treated with tegoprubart as part of immunosuppression regimen following islet transplantation in investigator-initiated trial at UChicago Medicine
Topline results from Phase 2 BESTOW trial of tegoprubart in kidney transplantation expected in fourth quarter of 2025
Proceeds from oversubscribed $85 million underwritten offering extend cash runway to end of 2026
IRVINE, Calif., March 20, 2025 (GLOBE NEWSWIRE) — Eledon Pharmaceuticals, Inc. (“Eledon”) (Nasdaq: ELDN) today reported its fourth quarter and full year 2024 operating and financial results and reviewed recent business highlights.
“We have recently made great strides in expanding our role in bringing new options in organ transplantation to patients. Tegoprubart was a cornerstone immunosuppression component in recent historic procedures including kidney xenotransplant and islet transplants in patients with type 1 diabetes,” said David-Alexandre C. Gros, M.D., Chief Executive Officer of Eledon. “The results from these landmark studies together with the encouraging allograft kidney transplant clinical data we have shared continue to reinforce tegoprubart’s broad potential to protect transplanted organs and cells, regardless of the transplant type and the organ source. We are entering 2025 from a position of balance sheet strength and we are on track to deliver on multiple key milestones in the coming months, including topline results from our Phase 2 BESTOW trial in the fourth quarter of 2025.”
Fourth Quarter 2024 and Recent Business Highlights
- Announced the use of tegoprubart as a lead component of the immunosuppression treatment regimen following the second transplant of a genetically modified pig kidney into a human in a study conducted at Massachusetts General Hospital. Following the successful transplant on January 25, 2025, the patient was discharged from the hospital without need for continued treatment with dialysis for the first time in over two years.
- Reported positive initial data for the first three islet transplant recipients treated with tegoprubart as part of an immunosuppression regimen for the prevention of islet transplant rejection in subjects with type 1 diabetes in an investigator-initiated trial at the University of Chicago Medicine’s Transplant Institute. The data demonstrated potentially the first in human cases of insulin independence achieved using an anti-CD40L monoclonal antibody immunosuppression therapy without the use of tacrolimus, the current standard for care for prevention of transplant rejection.
- Completed an oversubscribed, underwritten offering of common stock and pre-funded warrants for total gross proceeds of $85.0 million and net proceeds of approximately $79.5 million after deducting underwriting discounts, commissions, and offering expenses. The offering, which priced at a premium, included participation from both new and existing investors.
Anticipated Upcoming Milestones
- Summer 2025: Report updated interim clinical data from the ongoing Phase 1b open-label trial evaluating tegoprubart for the prevention of organ rejection in kidney transplant patients.
- 4Q 2025: Report topline results from the Phase 2 BESTOW trial of tegoprubart in kidney transplantation.
- 2025: Report updated interim clinical data from the investigator-led clinical trial with UChicago Medicine for pancreatic islet transplantation in subjects with type 1 diabetes.
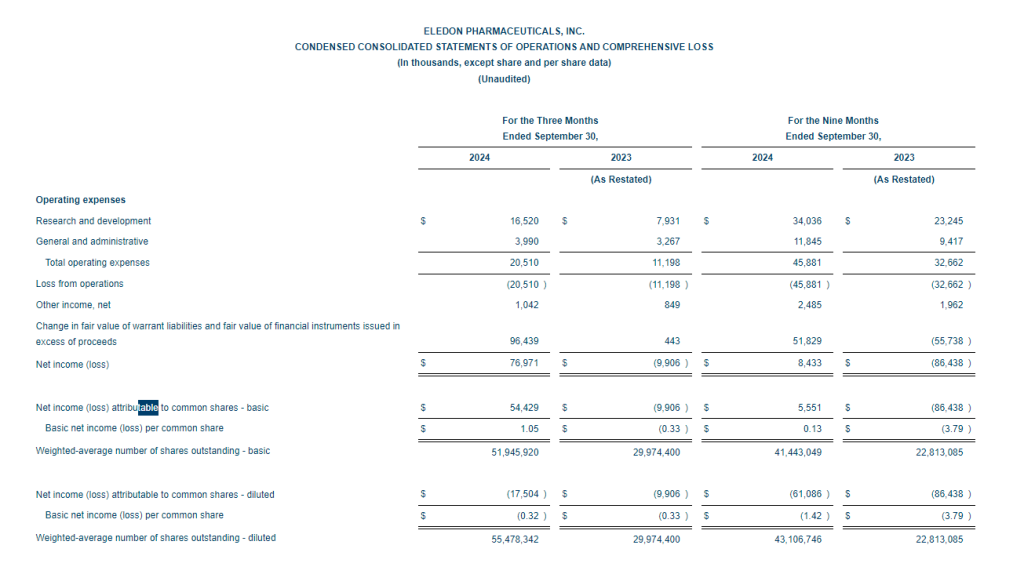
Fourth Quarter 2024 Financial Results
Cash, cash equivalents and short-term investments totaled $140.2 million as of December 31, 2024 compared to $78.2 million at September 30, 2024. The company expects current cash, cash equivalents and short-term investments to fund operations to the end of 2026.
Research and development (R&D) expenses for the fourth quarter of 2024 were $17.9 million, including $2.7 million of non-cash stock-based compensation expense, compared to $7.1 million, including $0.3 million of non-cash stock-based compensation expense, for the comparable period in 2023.
General and administrative expenses for the fourth quarter of 2024 were $6.8 million, including $3.9 million of non-cash stock-based compensation expense, compared to $3.3 million, including $1.4 million of non-cash stock-based compensation expense, for the comparable period in 2023.
Net loss for the fourth quarter of 2024 was $44.6 million, or $0.64 per basic share, compared with a net loss of $30.1 million, or $1.00 per basic share, for the comparable period in 2023. Both periods included a non-cash loss from changes in the fair value of warrant liabilities, totaling $20.9 million in 2024 and $20.5 million in 2023.
Full Year 2024 Financial Results
Research and development (R&D) expenses for the year ended December 31, 2024 were $52.0 million, including $4.3 million of non-cash stock-based compensation expense, compared to $30.3 million, including $1.5 million of non-cash stock-based compensation expense, for the comparable period in 2023. The increase was primarily driven by a rise in clinical development expenses related to the Phase 1b, Phase 2 BESTOW and Phase 2 open-label extension trials for kidney transplantation, an increase in manufacturing costs for drug substance and drug product clinical trial supply, an increase in stock-based compensation expense and employee compensation and benefits related to increased headcount.
General and administrative expenses for the year ended December 31, 2024 were $18.6 million, including $8.8 million of non-cash stock-based compensation expense, compared to $12.7 million, including $5.0 million of non-cash stock-based compensation expense, for the comparable period in 2023. The increase was primarily driven by an increase in stock-based compensation expense, professional services and employee compensation and benefits.
Net loss for the year ended December 31, 2024 was $36.2 million, or $0.75 per basic share, compared with a net loss of $116.5 million, or $4.73 per basic share, in 2023. The 2024 net loss included a non-cash gain of $30.9 million from changes in the fair value of warrant liabilities, whereas the 2023 net loss included a non-cash loss of $76.2 million from such changes. Excluding the non-cash items related to changes in the fair value of warrant liabilities, the company would have recorded a net loss of $67.1 million for the year ended December 31, 2024 and a net loss of $40.3 million for the year ended December 31, 2023.
About Eledon Pharmaceuticals and tegoprubart
Eledon Pharmaceuticals, Inc. is a clinical stage biotechnology company that is developing immune-modulating therapies for the management and treatment of life-threatening conditions. The Company’s lead investigational product is tegoprubart, an anti-CD40L antibody with high affinity for the CD40 Ligand, a well-validated biological target that has broad therapeutic potential. The central role of CD40L signaling in both adaptive and innate immune cell activation and function positions it as an attractive target for non-lymphocyte depleting, immunomodulatory therapeutic intervention. The Company is building upon a deep historical knowledge of anti-CD40 Ligand biology to conduct preclinical and clinical studies in kidney allograft transplantation, xenotransplantation, and amyotrophic lateral sclerosis (ALS). Eledon is headquartered in Irvine, California. For more information, please visit the Company’s website at www.eledon.com.
Follow Eledon Pharmaceuticals on social media: LinkedIn; Twitter
Forward Looking Statements
This press release contains forward-looking statements that involve substantial risks and uncertainties. Any statements about the company’s future expectations, plans and prospects, including statements about planned clinical trials, the development of product candidates, expected timing for initiation of future clinical trials, expected timing for receipt of data from clinical trials, the company’s capital resources and ability to finance planned clinical trials, as well as other statements containing the words “believes,” “anticipates,” “plans,” “expects,” “estimates,” “intends,” “predicts,” “projects,” “targets,” “looks forward,” “could,” “may,” and similar expressions, constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements are inherently uncertain and are subject to numerous risks and uncertainties, including: risks relating to the safety and efficacy of our drug candidates; risks relating to clinical development timelines, including interactions with regulators and clinical sites, as well as patient enrollment; risks relating to costs of clinical trials and the sufficiency of the company’s capital resources to fund planned clinical trials; and risks associated with the impact of the ongoing coronavirus pandemic. Actual results may differ materially from those indicated by such forward-looking statements as a result of various factors. These risks and uncertainties, as well as other risks and uncertainties that could cause the company’s actual results to differ significantly from the forward-looking statements contained herein, are discussed in our quarterly 10-Q, annual 10-K, and other filings with the U.S. Securities and Exchange Commission, which can be found at www.sec.gov. Any forward-looking statements contained in this press release speak only as of the date hereof and not of any future date, and the company expressly disclaims any intent to update any forward-looking statements, whether as a result of new information, future events or otherwise.
Investor Contact:
Stephen Jasper
Gilmartin Group
(858) 525 2047
stephen@gilmartinir.com
Media Contact:
Jenna Urban
CG Life
(212) 253 8881
jurban@cglife.com
Source: Eledon Pharmaceuticals
View Full Release Here.