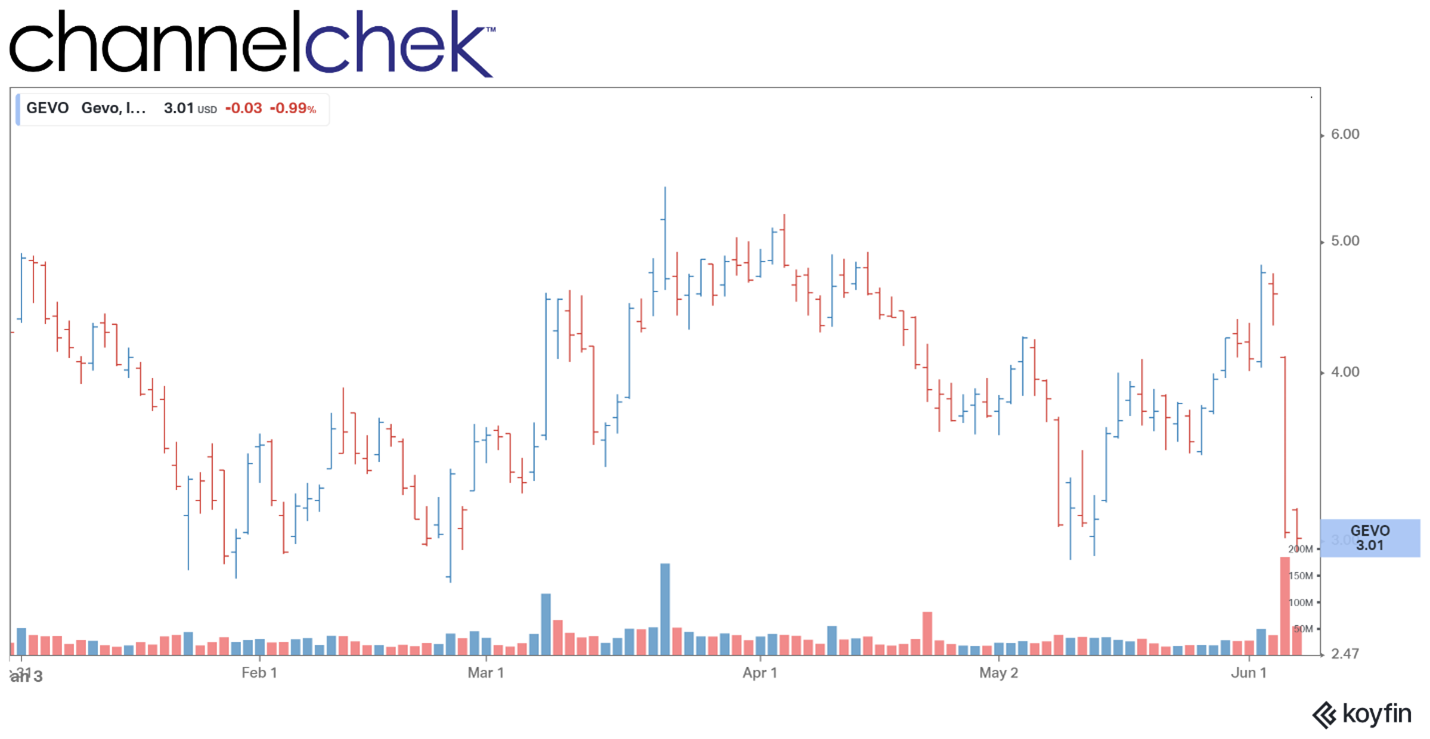
Ayala Pharmaceuticals Announces Data on AL101 in Adenoid Cystic Carcinoma (ACC) at 2022 ASCO Annual Meeting
Research, News, and Market Data on Ayala Pharmaceuticals
— Presentation at ASCO features updated
results from Phase 2 ACCURACY study —
— AL101 demonstrated anti-tumor activity by
achieving an overall disease control rate of 69% —
— Patients achieving partial responses had
higher progression-free survival —
REHOVOT, Israel & WILMINGTON, Del., June 06, 2022 (GLOBE NEWSWIRE) — Ayala Pharmaceuticals, Inc. (Nasdaq: AYLA), a clinical-stage oncology company focused on developing and commercializing small molecule therapeutics for patients suffering from rare and aggressive cancers, primarily in genetically defined patient populations, today is announcing data on AL101 from the Phase 2 ACCURACY study in a poster at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting taking place June 3-7, 2022 in Chicago, Illinois.
The poster at ASCO provides updated data from the 4mg and 6 mg AL101 cohorts in the ACCURACY study of the AL101, a selective gamma-secretase inhibitor, in subjects with recurrent/metastatic (R/M) adenoid cystic carcinoma (ACC) harboring Notch activating mutations (Notchmut) (NCT03691207). The most recent safety efficacy, PK, and PD data from the study is presented. The abstract from this study has been released on the 2022 ASCO Annual Meeting website (https://conferences.asco.org).
“We are excited to have AL101 featured in the 2022 ASCO meeting and are pleased with the very promising safety and efficacy data from ACCURACY,” said Gary Gordon, M.D., Ph.D., Chief Medical Officer of Ayala. “ACC is an orphan disease with no approved therapies and patients with Notch mutations have a more aggressive disease course and poorer survival outcomes as compared to patients with Notch wild-type. If approved, we see strong potential for AL101 to provide a much needed treatment option for R/M ACC patients and look forward to further advancing this clinical program.”
ACCURACY is an open-label, single-arm, multicenter Phase 2 study to assess the clinical activity of AL101 using radiographic assessments of patients with R/M ACC demonstrating disease progression within 6 months prior to dosing. It is the first ACC study selecting subjects with ACC bearing defined NOTCH-activating mutations. A total of 87 patients were enrolled (all with RECIST v1.1 evaluable disease or bone exclusive disease deemed evaluable by MD Anderson Bone Response criteria) and of these, 77 were evaluable for efficacy. Approximately 90% of the patients had metastatic disease at screening. This was a heavily pretreated patient population with over half having previously received some form of systemic therapy. Preliminary data from the study were announced by Ayala in 2021.
Efficacy Results
- In the 4mg dose group, 6 patients (14.6%) had a partial response, 23 (56.1%) had stable disease, for an overall disease control rate of 70.7%
- In the 6mg dose group, 3 patients (8.3%) had a partial response, 21 (58.3%) had stable disease and for an overall disease control rate of 66.7%
- Fifty six percent of the 4mg cohort patients and 36% of the 6mg cohort patients experienced some degree of tumor regression.
- The median progression free survival (PFS) in each of the 4mg and 6mg dose cohorts was 3.7 months but was 6.7 months among the patients who had a partial response
- Median overall survival (OS) was 9.3 months in the overall group but 12.1 months among the patients who had a partial response.
- Both dose regimens demonstrated substantial inhibition of the NOTCH pathway consistent with previous studies, but the higher dose did not improve the observed outcomes
Safety
- AL101 was adequately tolerated with most adverse events being Grade 1 or 2
- Among all 87 patients, 54 or 62% had treatment related grade 3 or 4 AEs (49% in the 4mg cohort and 76% in the 6mg cohort).
Dr. Renata Ferrarotto, Associate Professor and Director of Head and Neck Oncology Clinical Research at M.D. Anderson Cancer Center and the principal investigator of the study commented “The anti-tumor activity of AL101 in the ACCURACY study suggests that it is providing benefit to a subset of patients with ACC carrying Notch-activating mutations. We are presenting data for the first time showing that subjects who had a partial response to AL101 had progression free survival that was approximately double that of the group as a whole. This result, together with the high disease control rate observed, underscores the clinical relevance of the outcomes, given the aggressive disease course associated with Notch mutations and the fact that the majority of subjects in this trial were heavily pretreated.”
Poster presentation details:
|
Abstract Title:
|
|
Results of ACCURACY: A phase 2 trial of AL101, a selective gamma secretase inhibitor, in subjects with recurrent/metastatic (R/M) adenoid cystic carcinoma (ACC) harboring Notch activating mutations (Notchmut)
|
|
Abstract
Number:
|
|
6046
|
|
Session Title:
|
|
Head and Neck Cancer
|
|
Session
Date and Time:
|
|
Monday, June 6, 2022, 1:15 PM-4:15 PM CDT
|
A copy of the poster will be available on the Ayala corporate website, following the presentation at ASCO on June 6.
About Adenoid Cystic Carcinoma (ACC)
ACC is a rare malignancy of the secretory glands including salivary glands, accounting for about 10% of all salivary gland tumors with an annual incidence of 3,400 in the U.S. There is currently no approved standard of care for patients with recurrent/metastatic ACC. Patients with locoregional disease undergo surgery and radiation therapy, with recurring disease treated by chemotherapy. ACC is an immunologically “cold” tumor that is refractory to chemotherapy, with a recurrence rate of about 60% after initial surgery. The Notch pathway has been determined to be an oncogenic driver of ACC and its dysregulation plays a key role in tumorigenesis and correlates with a distinct pattern of metastasis and a poor prognosis.
About AL101
AL101 is an investigational small molecule Gamma Secretase Inhibitor (GSI) that is designed to potently and selectively inhibit Notch 1, 2, 3 and 4, and is currently being evaluated in the Phase 2 ACCURACY clinical studies in patients with adenoid cystic carcinoma (ACC). AL101 is designed to inhibit the expression of Notch gene targets by blocking the final cleavage step by the gamma secretase required for Notch activation. Ayala obtained an exclusive, worldwide license to develop and commercialize AL101 from Bristol-Myers Squibb Company in November 2017. AL101 was granted U.S. FDA Fast Track Designation and Orphan Drug Designation for the treatment of ACC.
About Ayala Pharmaceuticals
Ayala Pharmaceuticals, Inc. is a clinical-stage oncology company focused on developing and commercializing small molecule therapeutics for patients suffering from rare and aggressive cancers, primarily in genetically defined patient populations. Ayala’s approach is focused on predicating, identifying and addressing tumorigenic drivers of cancer through a combination of its bioinformatics platform and next-generation sequencing to deliver targeted therapies to underserved patient populations. The company has two product candidates under development, AL101 and AL102, targeting the aberrant activation of the Notch pathway with gamma secretase inhibitors to treat a variety of tumors including Desmoid Tumors, Adenoid Cystic Carcinoma and T-cell Acute Lymphoblastic Leukemia (T-ALL). AL101, has received Fast Track Designation and Orphan Drug Designation from the U.S. FDA and is currently in a Phase 2 clinical trial for patients with ACC (ACCURACY) bearing Notch activating mutations. AL102 is currently in a Pivotal Phase 2/3 clinical trials for patients with desmoid tumors (RINGSIDE). For more information, visit www.ayalapharma.com.
Contacts:
Investors:
Joyce Allaire
LifeSci Advisors LLC
+1-617-435-6602
jallaire@lifesciadvisors.com
Ayala Pharmaceuticals:
+1-857-444-0553
info@ayalapharma.com
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including statements relating to our development of AL101 and AL102, the promise and potential impact of our preclinical or clinical trial data, the timing of and plans to initiate additional clinical trials of AL101 and AL102, the timing and results of any clinical trials or readouts, the sufficiency of cash to fund operations, and the anticipated impact of COVID-19, on our business. These forward-looking statements are based on management’s current expectations. The words ”may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “estimate,” “believe,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words.
These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: we have incurred significant losses since inception and anticipate that we will continue to incur losses for the foreseeable future. We are not currently profitable, and we may never achieve or sustain profitability; we will require additional capital to fund our operations, and if we fail to obtain necessary financing, we may not be able to complete the development and commercialization of AL101 and AL102; we have a limited operating history and no history of commercializing pharmaceutical products, which may make it difficult to evaluate the prospects for our future viability; we are heavily dependent on the success of AL101 and AL102, our most advanced product candidates, which are still under clinical development, and if either AL101 or AL102 does not receive regulatory approval or is not successfully commercialized, our business may be harmed; due to our limited resources and access to capital, we must prioritize development of certain programs and product candidates; these decisions may prove to be wrong and may adversely affect our business; the outbreak of COVID-19, may adversely affect our business, including our clinical trials; our ability to use our net operating loss carry forwards to offset future taxable income may be subject to certain limitations; our product candidates are designed for patients with genetically defined cancers, which is a rapidly evolving area of science, and the approach we are taking to discover and develop product candidates is novel and may never lead to marketable products; we were not involved in the early development of our lead product candidates; therefore, we are dependent on third parties having accurately generated, collected and interpreted data from certain preclinical studies and clinical trials for our product candidates; enrollment and retention of patients in clinical trials is an expensive and time-consuming process and could be made more difficult or rendered impossible by multiple factors outside our control; if we do not achieve our projected development and commercialization goals in the timeframes we announce and expect, the commercialization of our product candidates may be delayed and our business will be harmed; our product candidates may cause serious adverse events or undesirable side effects, which may delay or prevent marketing approval, or, if approved, require them to be taken off the market, require them to include safety warnings or otherwise limit their sales; the market opportunities for AL101 and AL102, if approved, may be smaller than we anticipate; we may not be successful in developing, or collaborating with others to develop, diagnostic tests to identify patients with Notch-activating mutations; we have never obtained marketing approval for a product candidate and we may be unable to obtain, or may be delayed in obtaining, marketing approval for any of our product candidates; even if we obtain FDA approval for our product candidates in the United States, we may never obtain approval for or commercialize them in any other jurisdiction, which would limit our ability to realize their full market potential; we have been granted Orphan Drug Designation for AL101 for the treatment of ACC and may seek Orphan Drug Designation for other indications or product candidates, and we may be unable to maintain the benefits associated with Orphan Drug Designation, including the potential for market exclusivity, and may not receive Orphan Drug Designation for other indications or for our other product candidates; although we have received Fast Track designation for AL101, and may seek Fast Track designation for our other product candidates, such designations may not actually lead to a faster development timeline, regulatory review or approval process; we face significant competition from other biotechnology and pharmaceutical companies and our operating results will suffer if we fail to compete effectively; we are dependent on a small number of suppliers for some of the materials used to manufacture our product candidates, and on one company for the manufacture of the active pharmaceutical ingredient for each of our product candidates; any future collaborations will be, important to our business. If we are unable to maintain our existing collaboration or enter into new collaborations, or if these collaborations are not successful, our business could be adversely affected; enacted and future healthcare legislation may increase the difficulty and cost for us to obtain marketing approval of and commercialize our product candidates, if approved, and may affect the prices we may set; if we are unable to obtain, maintain, protect and enforce patent and other intellectual property protection for our technology and products or if the scope of the patent or other intellectual property protection obtained is not sufficiently broad, our competitors could develop and commercialize products and technology similar or identical to ours, and we may not be able to compete effectively in our markets; we may engage in acquisitions or in-licensing transactions that could disrupt our business, cause dilution to our stockholders or reduce our financial resources; and risks related to our operations in Israel could materially adversely impact our business, financial condition and results of operations.
These and other important factors discussed under the caption “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2021 filed with the U.S. Securities and Exchange Commission (SEC) on March 28, 2022 and our other filings with the SEC, could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management’s estimates as of the date of this press release. New risk factors and uncertainties may emerge from time to time, and it is not possible to predict all risk factors and uncertainties. While we may elect to update such forward-looking statements at some point in the future, except as required by law, we disclaim any obligation to do so, even if subsequent events cause our views to change. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this press release.