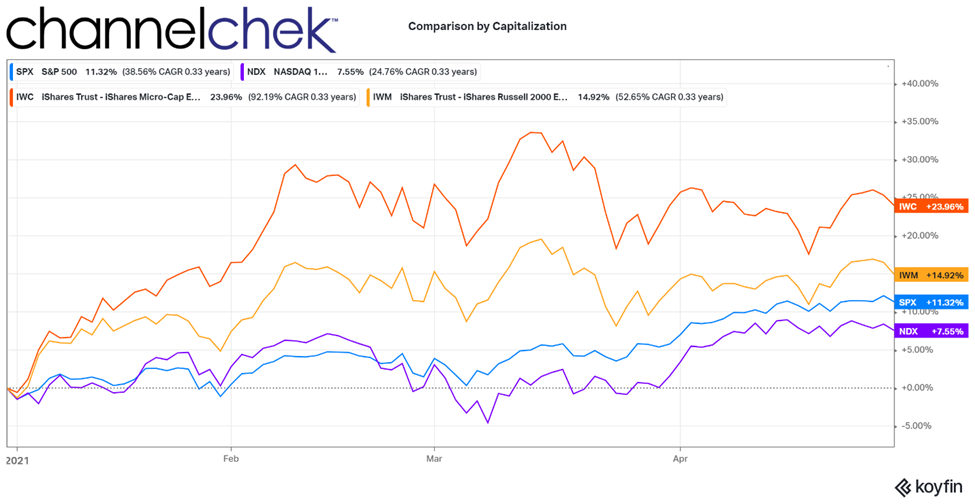
OpRegen® Clinical Data Continues To Demonstrate Improvements In Patients With Dry AMD With Geographic Atrophy
- Eighty-Three Percent of All Cohort 4 Patients Exhibited Stable or Improved BCVA
- Visual Acuity Declined in the Majority of Untreated Eyes
- Positive Interim Outcomes on Patient-Reported Visual Function Questionnaire
- No Acute or Delayed Inflammation or Rejection Events, Even in Patients Receiving Reduced Immunosuppressive Regimen
- Previously Reported Evidence of Retinal Restoration Has Persisted to Month 35
- Data Reported at 2021 ARVO Meeting
CARLSBAD, Calif.–(BUSINESS WIRE)–May 3, 2021–
Lineage Cell Therapeutics, Inc.
(NYSE American and TASE: LCTX), a clinical-stage biotechnology company developing novel cell therapies for unmet medical needs, announced today that updated interim results from its ongoing, 24-patient Phase 1/2a clinical study of its lead product candidate, OpRegen, were reported at the 2021 Association for Research in Vision and Ophthalmology Annual Meeting (ARVO 2021). OpRegen is an investigational cell therapy consisting of allogeneic retinal pigment epithelium (RPE) cells administered to the subretinal space for the treatment of dry age-related macular degeneration (AMD) with geographic atrophy (GA). At ARVO 2021, additional data were presented on 24 patients enrolled in the study, including all 12 patients treated in Cohort 4, which have better baseline vision and smaller areas of GA than earlier cohorts.
“I continue to be very excited about this work and the clinical data generated to date with OpRegen, especially in the better vision Cohort 4 patients,” stated Principal Investigator
Christopher D. Riemann, M.D., Vitreoretinal Surgeon and Fellowship Director,
Cincinnati Eye Institute and
University of Cincinnati School of Medicine. “There seems to be a significant visual acuity signal in Cohort 4 patients, with most treated eyes having stable or improved vision over time when compared to the contralateral eyes having stable or worsening vision over time. When looking at reading speed progression, treated eyes also seemed to improve while untreated eyes declined. Notably, some individual responders had impressive visual acuity improvements and reductions in GA progression compared to their contralateral eyes. Most importantly, we believe that earlier intervention in less severely affected patients along with a more central placement of transplanted OpRegen cells may increase the likelihood of a clinically beneficial effect. Overall, these results are encouraging and are of a magnitude that could be clinically very important if confirmed in further clinical studies.”
“These new data continue to indicate to us that treatment with OpRegen can generate clinically meaningful outcomes in dry AMD patients with GA, particularly in those with earlier-stage disease,” stated
Brian M. Culley, Lineage CEO. “It also appears that earlier intervention in less severely affected patients and more central placement of the transplanted cells may increase the likelihood of observing a benefit. Additionally, among the newly reported data, it was notable that Cohort 4 patients reported improvements in a majority of the vision parameters measured by a validated quality of life questionnaire. The magnitude of these improvements was higher overall in Cohort 4 than in Cohorts 1 through 3, which is consistent with the larger clinical benefit observed among those patients. As this data set matures, our efforts turn next to evaluating the six most recently treated Cohort 4 patients for indications of retinal restoration and reductions in the size and growth of the areas of GA. Our overall objective is to position OpRegen RPE transplants as the clear leader in the race to address the large unmet need in dry AMD with GA and establish Lineage as the pre-eminent allogeneic cell therapy company.”
Updated results presented at ARVO 2021 included a minimum of 4.5 months of follow-up in all 24 patients treated with OpRegen. Nine of twenty-four patients were treated with the “thaw and inject” formulation of OpRegen, two via a standard pars plana vitrectomy (PPV) and seven utilizing the Orbit™ Subretinal Delivery System (Orbit SDS).
Overall, 10/12 (83%) of the Cohort 4 patients’ treated eyes were at or above baseline visual acuity at their last assessment, based on per protocol scheduled visits ranging from 4.5 months to approximately 3 years post-transplant. Improvements in best corrected visual acuity (BCVA) for Cohort 4 patients reached up to +19 letters on the Early Treatment Diabetic Retinopathy Study (ETDRS) chart. In contrast, 10/12 (83%) of the patients’ untreated eyes were below pre-treatment baseline values at the same time points. Among the newly reported data, three (50%) of the more recently treated Cohort 4 patients exhibited marked improvements in BCVA ranging from +7 to +16 letters at their last scheduled assessments of at least 4.5 months. Two additional Cohort 4 patients experienced a gain of 2 letters from their baseline values. One Cohort 4 patient measured 7 letters below baseline. Previously reported structural improvements in the retina, decreases in drusen density, and a trend toward slower GA progression in treated compared to untreated eyes continued. Overall, OpRegen has been well tolerated with no unexpected adverse events or serious adverse events, and evidence of durable engraftment of OpRegen RPE cells have extended to more than 5 years in earliest treated patients, supporting the potential for OpRegen to be a one-time treatment.
A Cohort 4 patient with evidence of retinal restoration and confirmed history of GA growth, which was first reported 9 months following treatment, continues to demonstrate areas of retinal restoration as of their last assessment, approximately 3 years after treatment.
2021 ARVO Presentation OpRegen Data Highlights (As of April 16, 2021):
- In Cohort 1-3 patients (all legally blind at baseline), visual acuity reductions occurred as expected due to progressive GA;
- In Cohort 4 patients, which collectively had smaller areas of GA and higher baseline BCVA as compared to Cohort 1-3 patients, improved or sustained BCVA has been observed in 10/12 (83%) patients as of their last visit prior to this update (range of -7 to +19 letters on the ETDRS chart);
- OpRegen continues to be well-tolerated in all treated patients (N = 24);
- The majority of adverse events were mild (87%);
- Sustained subretinal pigmentation continues to suggest multi-year durability of OpRegen transplants;
- Improved anatomy and function continue to be observed in some patients, including:
- Reduction in drusen;
- Photoreceptor and RPE layer restoration;
- Localized slowing of GA progression in treated areas;
- Better visual acuity via ETDRS scores and reading speed; and
- Improved National Eye Institute Visual Function Questionnaire (VFQ-25) scores.
- Post-treatment surgical interventions occurred in four cases (5 events in 4 patients):
- Three epiretinal membranes (ERM) were surgically peeled. Mild to moderate ERM were observed in an additional 12 out of 17 PPV operated patients. Most ERMs were clinically insignificant.
- Retinal detachment (RD) was observed in 2 out of 17 patients, neither of which appears to be attributable to OpRegen or any study related medications:
- The first case of RD, which occurred in a Cohort 3 patient, was an unsuccessful repair of a post-surgical retinal tear; visual acuity did not regain baseline levels; and
- The second case of RD, which occurred in a Cohort 4 patient, was successfully repaired; post-surgical visual acuity has remained higher than baseline.
- Choroidal neovascularization (CNV) was observed in 3 out of 7 patients receiving OpRegen via the Orbit SDS, all of whom received treatment with an approved anti-VEGF;
- As previously reported, one PPV operated patient developed CNV, which was identified more than two years following treatment.
As part of an ongoing effort to administer the minimally effective dose and duration of immunosuppressive therapy, immunosuppression was utilized only during the perioperative period of approximately 3 months in Cohort 4 patients. One patient received a modified immunosuppressive regimen at baseline, which included no tacrolimus and only mycophenolate mofetil. One patient was diagnosed with COVID-19 shortly after treatment for whom all immunosuppression was halted and reinstated once the patient was asymptomatic. Both patients showed no signs of acute or delayed inflammation or rejection of OpRegen cells with 4.5 months of post-transplant follow up. Other than the reduced regimens described above, immunosuppressants have been discontinued as scheduled, typically within 90 days post-operatively, and no cases of acute or delayed rejection or inflammation due to OpRegen have been reported in any patients treated with OpRegen.
The presentation, “Phase I/IIa Clinical Trial of Transplanted Allogeneic Retinal Pigmented Epithelium (RPE, OpRegen) Cells in Advanced Dry Age-Related Macular Degeneration (AMD): Interim Results” is being featured as part of the Stem Cells/Gene Therapy/Transplantation Session, on
May 6, 2021
between 5:15 pm and 6:45 pm EDT by
Christopher D. Riemann, M.D.(abstract number 3538173).
About OpRegen
OpRegen is currently being evaluated in a Phase 1/2a open-label, dose escalation safety and efficacy study of a single injection of human retinal pigment epithelium cells derived from an established pluripotent cell line and transplanted subretinally in patients with advanced dry AMD with GA. The study enrolled 24 patients into 4 cohorts. The first 3 cohorts enrolled only legally blind patients with BCVA of 20/200 or worse. The fourth cohort enrolled 12 better vision patients (vision from 20/65 to 20/250 with smaller areas of GA). Cohort 4 also included patients treated with a new “thaw-and-inject” formulation of OpRegen, which can be shipped directly to sites and used immediately upon thawing, removing the complications and logistics of having to use a dose preparation facility. The primary objective of the study is to evaluate the safety and tolerability of OpRegen as assessed by the incidence and frequency of treatment emergent adverse events. Secondary objectives are to evaluate the preliminary efficacy of OpRegen treatment by assessing the changes in ophthalmological parameters measured by various methods of primary clinical relevance. Additional objectives include the evaluation of the safety of delivery of OpRegen using the Orbit SDS. OpRegen is a registered trademark of
Cell Cure Neurosciences Ltd., a majority-owned subsidiary of
Lineage Cell Therapeutics, Inc. The Orbit subretinal delivery system is used under agreement with
Gyroscope Therapeutics Limited. Orbit and Orbit SDS are trademarks of
Gyroscope Therapeutics Limited.
About Dry AMD
Dry age-related macular degeneration (AMD) is a leading cause of adult blindness in the developed world. There are two forms of AMD: wet AMD and dry AMD. Dry AMD is the more common of the two types, accounting for approximately 85-90% of cases. Wet AMD is the less common of the two types, accounting for approximately 10-15% of cases. Global sales of the two leading wet AMD therapies were in excess of
$10 billion in 2019. Nearly all cases of wet AMD begin as dry AMD. Dry AMD typically affects both eyes. There are currently no
U.S. Food and Drug Administration or
European Medicines Agency approved treatment options available for patients with dry AMD.
About Lineage Cell Therapeutics, Inc.
Lineage Cell Therapeutics is a clinical-stage biotechnology company developing novel cell therapies for unmet medical needs. Lineage’s programs are based on its robust proprietary cell-based therapy platform and associated in-house development and manufacturing capabilities. With this platform Lineage develops and manufactures specialized, terminally differentiated human cells from its pluripotent and progenitor cell starting materials. These differentiated cells are developed to either replace or support cells that are dysfunctional or absent due to degenerative disease or traumatic injury or administered as a means of helping the body mount an effective immune response to cancer. Lineage’s clinical programs are in markets with billion dollar opportunities and include three allogeneic (“off-the-shelf”) product candidates: (i) OpRegen®, a retinal pigment epithelium transplant therapy in Phase 1/2a development for the treatment of dry age-related macular degeneration, a leading cause of blindness in the developed world; (ii) OPC1, an oligodendrocyte progenitor cell therapy in Phase 1/2a development for the treatment of acute spinal cord injuries; and (iii) VAC, an allogeneic dendritic cell therapy platform for immuno-oncology and infectious disease, currently in clinical development for the treatment of non-small cell lung cancer. For more information, please visit www.lineagecell.com or follow the Company on Twitter @LineageCell.
Forward-Looking Statements
Lineage cautions you that all statements, other than statements of historical facts, contained in this press release, are forward-looking statements. Forward-looking statements, in some cases, can be identified by terms such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “design,” “intend,” “expect,” “could,” “plan,” “potential,” “predict,” “seek,” “should,” “would,” “contemplate,” project,” “target,” “tend to,” or the negative version of these words and similar expressions. Such statements include, but are not limited to, statements relating to the expected clinical outcomes of treatment with OpRegen in dry AMD patients with GA. Forward-looking statements involve known and unknown risks, uncertainties and other factors that may cause Lineage’s actual results, performance or achievements to be materially different from future results, performance or achievements expressed or implied by the forward-looking statements in this press release, including risks and uncertainties inherent in Lineage’s business and other risks in Lineage’s filings with the
Securities and Exchange Commission (SEC). Lineage’s forward-looking statements are based upon its current expectations and involve assumptions that may never materialize or may prove to be incorrect. All forward-looking statements are expressly qualified in their entirety by these cautionary statements. Further information regarding these and other risks is included under the heading “Risk Factors” in Lineage’s periodic reports with the
SEC, including Lineage’s most recent Annual Report on Form 10-K filed with the
SEC and its other reports, which are available from the SEC’s website. You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date on which they were made. Lineage undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
Lineage Cell Therapeutics, Inc. IR
Ioana C. Hone
(ir@lineagecell.com)
(442) 287-8963
Solebury Trout IR
Gitanjali Jain Ogawa
(Gogawa@soleburytrout.com)
(646) 378-2949
Russo Partners – Media Relations
Nic Johnson or
David Schull
Nic.johnson@russopartnersllc.com
David.schull@russopartnersllc.com
(212) 845-4242
Source:
Lineage Cell Therapeutics, Inc.